| Record Information |
|---|
| Version | 5.0 |
|---|
| Status | Detected and Quantified |
|---|
| Creation Date | 2006-08-13 15:33:17 UTC |
|---|
| Update Date | 2022-03-07 02:49:20 UTC |
|---|
| HMDB ID | HMDB0004264 |
|---|
| Secondary Accession Numbers | - HMDB0011138
- HMDB04264
- HMDB11138
|
|---|
| Metabolite Identification |
|---|
| Common Name | 14,15-Epoxy-5,8,11-eicosatrienoic acid |
|---|
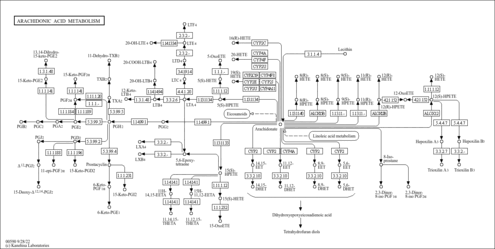
| Description | 14,15-epoxy-5,8,11-eicosatrienoic acid is an epoxyeicosatrienoic acid (EET), a metabolite of arachidonic acid. The P450 eicosanoids epoxyeicosatrienoic acids (EETs) are endogenous lipid mediators produced by P450 epoxygenases and metabolized through multiple pathways including soluble epoxide hydrolase (sEH). The cytochrome P-450 (P450) monooxygenase pathway includes enzymes of the CYP1A, CYP2B, CYP2C, CYP2E, and CYP2J subfamilies that catalyze the formation of four regioisomeric products, 5,6-, 8,9-, 11,12-, and 14,15-epoxyeicosatrienoic acid. EETs are produced in brain and perform important biological functions, including protection from ischemic injury. Both light flashes and direct glial stimulation produce vasodilatation mediated by EETs. EETs may be involved in the development of hypertension and endothelial dysfunction in DOCA-salt rats, but not in excessive collagen deposition or electrophysiological abnormalities. EETs have vasodilator and natriuretic effect. Blockade of EET formation is associated with salt-sensitive hypertension. Four regioisomeric cis-EET are primary products of arachidonic acid metabolism by cytochrome P450 epoxygenases. Upon hydration by soluble epoxide hydrolase (sEH), EET are metabolized to dihydroxyeicosatrienoic acids (DHET). These hydration products are more stable and less biologically active than EETs. (PMID: 17494091 , 17468203 , 17434916 , 17406062 , 17361113 , 15581597 ). |
|---|
| Structure | CCCCCC1OC1C\C=C/C\C=C/C\C=C/CCCC(O)=O InChI=1S/C20H32O3/c1-2-3-12-15-18-19(23-18)16-13-10-8-6-4-5-7-9-11-14-17-20(21)22/h4,6-7,9-10,13,18-19H,2-3,5,8,11-12,14-17H2,1H3,(H,21,22)/b6-4-,9-7-,13-10- |
|---|
| Synonyms | | Value | Source |
|---|
| (+/-)14,15-epetre | ChEBI | | (5Z,8Z,11Z)-14,15-Epoxyeicosa-5.8.11-trienoic acid | ChEBI | | (5Z,8Z,11Z)-14,15-Epoxyicosa-5.8.11-trienoic acid | ChEBI | | 14(15)-EpETrE | ChEBI | | 14,15-EpETrE | ChEBI | | 14,15-Epoxy-5Z,8Z,11Z-eicosatrienoic acid | ChEBI | | 14,15-Epoxy-5Z,8Z,11Z-icosatrienoic acid | ChEBI | | all-cis-14,15-Epoxyeicosa-5,8,11-trienoic acid | ChEBI | | (5Z,8Z,11Z)-14,15-Epoxyeicosa-5.8.11-trienoate | Generator | | (5Z,8Z,11Z)-14,15-Epoxyicosa-5.8.11-trienoate | Generator | | 14,15-Epoxy-5Z,8Z,11Z-eicosatrienoate | Generator | | 14,15-Epoxy-5Z,8Z,11Z-icosatrienoate | Generator | | all-cis-14,15-Epoxyeicosa-5,8,11-trienoate | Generator | | 14,15-Epoxy-5,8,11-eicosatrienoate | Generator | | 14(15)-EpETrE-ea | HMDB | | 14,15-EET | HMDB | | 14,15-Epoxy-5,8,11-eicosatrienoic acid, (2alpha(5Z,8Z,11Z),3alpha)-isomer | HMDB | | 14,15-Epoxyeicosatrienoic acid | HMDB | | 14,15-Oxido-5,8,11-eicosatrienoic acid | HMDB |
|
|---|
| Chemical Formula | C20H32O3 |
|---|
| Average Molecular Weight | 320.4663 |
|---|
| Monoisotopic Molecular Weight | 320.23514489 |
|---|
| IUPAC Name | (5Z,8Z,11Z)-13-(3-pentyloxiran-2-yl)trideca-5,8,11-trienoic acid |
|---|
| Traditional Name | 14,15-epoxyeicosatrienoic acid |
|---|
| CAS Registry Number | 81276-03-1 |
|---|
| SMILES | CCCCCC1OC1C\C=C/C\C=C/C\C=C/CCCC(O)=O |
|---|
| InChI Identifier | InChI=1S/C20H32O3/c1-2-3-12-15-18-19(23-18)16-13-10-8-6-4-5-7-9-11-14-17-20(21)22/h4,6-7,9-10,13,18-19H,2-3,5,8,11-12,14-17H2,1H3,(H,21,22)/b6-4-,9-7-,13-10- |
|---|
| InChI Key | JBSCUHKPLGKXKH-ILYOTBPNSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | Belongs to the class of organic compounds known as long-chain fatty acids. These are fatty acids with an aliphatic tail that contains between 13 and 21 carbon atoms. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Lipids and lipid-like molecules |
|---|
| Class | Fatty Acyls |
|---|
| Sub Class | Fatty acids and conjugates |
|---|
| Direct Parent | Long-chain fatty acids |
|---|
| Alternative Parents | |
|---|
| Substituents | - Long-chain fatty acid
- Epoxy fatty acid
- Heterocyclic fatty acid
- Unsaturated fatty acid
- Carboxylic acid derivative
- Carboxylic acid
- Dialkyl ether
- Oxirane
- Ether
- Monocarboxylic acid or derivatives
- Organoheterocyclic compound
- Oxacycle
- Organic oxide
- Hydrocarbon derivative
- Carbonyl group
- Organooxygen compound
- Organic oxygen compound
- Aliphatic heteromonocyclic compound
|
|---|
| Molecular Framework | Aliphatic heteromonocyclic compounds |
|---|
| External Descriptors | |
|---|
| Ontology |
|---|
| Physiological effect | Not Available |
|---|
| Disposition | |
|---|
| Process | |
|---|
| Role | Not Available |
|---|
| Physical Properties |
|---|
| State | Solid |
|---|
| Experimental Molecular Properties | | Property | Value | Reference |
|---|
| Melting Point | Not Available | Not Available | | Boiling Point | Not Available | Not Available | | Water Solubility | Not Available | Not Available | | LogP | Not Available | Not Available |
|
|---|
| Experimental Chromatographic Properties | Experimental Collision Cross Sections |
|---|
| Predicted Molecular Properties | |
|---|
| Predicted Chromatographic Properties | Predicted Collision Cross SectionsPredicted Kovats Retention IndicesUnderivatizedDerivatized| Derivative Name / Structure | SMILES | Kovats RI Value | Column Type | Reference |
|---|
| 14,15-Epoxy-5,8,11-eicosatrienoic acid,1TMS,isomer #1 | CCCCCC1OC1C/C=C\C/C=C\C/C=C\CCCC(=O)O[Si](C)(C)C | 2465.1 | Semi standard non polar | 33892256 | | 14,15-Epoxy-5,8,11-eicosatrienoic acid,1TBDMS,isomer #1 | CCCCCC1OC1C/C=C\C/C=C\C/C=C\CCCC(=O)O[Si](C)(C)C(C)(C)C | 2716.6 | Semi standard non polar | 33892256 |
|
|---|
| GC-MS Spectra| Spectrum Type | Description | Splash Key | Deposition Date | Source | View |
|---|
| Predicted GC-MS | Predicted GC-MS Spectrum - 14,15-Epoxy-5,8,11-eicosatrienoic acid GC-MS (Non-derivatized) - 70eV, Positive | splash10-0a4j-7390000000-0a6ea9221818f7ca6b61 | 2017-09-01 | Wishart Lab | View Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - 14,15-Epoxy-5,8,11-eicosatrienoic acid GC-MS (1 TMS) - 70eV, Positive | splash10-00bj-9152000000-75e265bfced30aca2f55 | 2017-10-06 | Wishart Lab | View Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - 14,15-Epoxy-5,8,11-eicosatrienoic acid GC-MS (Non-derivatized) - 70eV, Positive | Not Available | 2021-10-12 | Wishart Lab | View Spectrum |
MS/MS Spectra| Spectrum Type | Description | Splash Key | Deposition Date | Source | View |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 14,15-Epoxy-5,8,11-eicosatrienoic acid 10V, Positive-QTOF | splash10-0udi-1139000000-d1b1217f1a0ab449ee76 | 2017-09-01 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 14,15-Epoxy-5,8,11-eicosatrienoic acid 20V, Positive-QTOF | splash10-0f80-9562000000-3a641d61ec600d27001a | 2017-09-01 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 14,15-Epoxy-5,8,11-eicosatrienoic acid 40V, Positive-QTOF | splash10-0f77-9610000000-4d2f2f7c7445ce34caa9 | 2017-09-01 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 14,15-Epoxy-5,8,11-eicosatrienoic acid 10V, Negative-QTOF | splash10-014i-1029000000-03d83d003242098cb3c2 | 2017-09-01 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 14,15-Epoxy-5,8,11-eicosatrienoic acid 20V, Negative-QTOF | splash10-0gb9-4159000000-4a32a74e26b6a8978859 | 2017-09-01 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 14,15-Epoxy-5,8,11-eicosatrienoic acid 40V, Negative-QTOF | splash10-052f-9120000000-efc218792d62c8df73a5 | 2017-09-01 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 14,15-Epoxy-5,8,11-eicosatrienoic acid 10V, Positive-QTOF | splash10-0uki-2259000000-adfdaf1e87da0a4fbf5f | 2021-09-23 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 14,15-Epoxy-5,8,11-eicosatrienoic acid 20V, Positive-QTOF | splash10-0udr-9375000000-fdf50048f25a924a6bb2 | 2021-09-23 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 14,15-Epoxy-5,8,11-eicosatrienoic acid 40V, Positive-QTOF | splash10-00mo-9200000000-f15fc52cfe999f85655e | 2021-09-23 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 14,15-Epoxy-5,8,11-eicosatrienoic acid 10V, Negative-QTOF | splash10-014i-0009000000-fa339ab07ef5a66d93e2 | 2021-09-25 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 14,15-Epoxy-5,8,11-eicosatrienoic acid 20V, Negative-QTOF | splash10-0gb9-2149000000-f932d9bc138901e04dad | 2021-09-25 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 14,15-Epoxy-5,8,11-eicosatrienoic acid 40V, Negative-QTOF | splash10-052f-9140000000-91f3f678f570fc1a1ff6 | 2021-09-25 | Wishart Lab | View Spectrum |
|
|---|