You are using an unsupported browser. Please upgrade your browser to a newer version to get the best experience on Human Metabolome Database.
| Record Information |
|---|
| Version | 5.0 |
|---|
| Status | Detected and Quantified |
|---|
| Creation Date | 2005-11-16 15:48:42 UTC |
|---|
| Update Date | 2022-03-07 02:49:03 UTC |
|---|
| HMDB ID | HMDB0000561 |
|---|
| Secondary Accession Numbers | |
|---|
| Metabolite Identification |
|---|
| Common Name | beta-Carotene |
|---|
| Description | Beta-Carotene belongs to the class of organic compounds known as carotenes. These are a type of polyunsaturated hydrocarbon molecules containing eight consecutive isoprene units. Carotenes are characterized by the presence of two end-groups (mostly cyclohexene rings, but also cyclopentene rings or acyclic groups) linked by a long branched alkyl chain. Beta-carotene is therefore considered to be an isoprenoid lipid molecule. Beta-carotene is a strongly coloured red-orange pigment abundant in fungi, plants, and fruits. It is synthesized biochemically from eight isoprene units and therefore has 40 carbons. Among the carotenes, beta-carotene is distinguished by having beta-rings at both ends of the molecule. Beta-Carotene is biosynthesized from geranylgeranyl pyrophosphate. It is the most common form of carotene in plants. In nature, Beta-carotene is a precursor (inactive form) to vitamin A. Vitamin A is produed via the action of beta-carotene 15,15'-monooxygenase on carotenes. In mammals, carotenoid absorption is restricted to the duodenum of the small intestine and dependent on a class B scavenger receptor (SR-B1) membrane protein, which is also responsible for the absorption of vitamin E. One molecule of beta-carotene can be cleaved by the intestinal enzyme Beta-Beta-carotene 15,15'-monooxygenase into two molecules of vitamin A. Beta-Carotene contributes to the orange color of many different fruits and vegetables. Vietnamese gac and crude palm oil are particularly rich sources, as are yellow and orange fruits, such as cantaloupe, mangoes, pumpkin, and papayas, and orange root vegetables such as carrots and sweet potatoes. Excess beta-carotene is predominantly stored in the fat tissues of the body. The most common side effect of excessive beta-carotene consumption is carotenodermia, a physically harmless condition that presents as a conspicuous orange skin tint arising from deposition of the carotenoid in the outermost layer of the epidermis. |
|---|
| Structure | C\C(\C=C\C=C(/C)\C=C\C1=C(C)CCCC1(C)C)=C/C=C/C=C(\C)/C=C/C=C(\C)/C=C/C1=C(C)CCCC1(C)C InChI=1S/C40H56/c1-31(19-13-21-33(3)25-27-37-35(5)23-15-29-39(37,7)8)17-11-12-18-32(2)20-14-22-34(4)26-28-38-36(6)24-16-30-40(38,9)10/h11-14,17-22,25-28H,15-16,23-24,29-30H2,1-10H3/b12-11+,19-13+,20-14+,27-25+,28-26+,31-17+,32-18+,33-21+,34-22+ |
|---|
| Synonyms | | Value | Source |
|---|
| 1,1'-[(1E,3E,5E,7E,9E,11E,13E,15E,17E)-3,7,12,16-Tetramethyloctadeca-1,3,5,7,9,11,13,15,17-nonaene-1,18-diyl]bis(2,6,6-trimethylcyclohexene) | ChEBI | | all-trans-beta-Carotene | ChEBI | | beta-Karotin | ChEBI | | all-trans-b-Carotene | Generator | | all-trans-Β-carotene | Generator | | b-Karotin | Generator | | Β-karotin | Generator | | b-Carotene | Generator | | Β-carotene | Generator | | (all-e)-1,1'-(3,7,12,16-Tetramethyl-1,3,5,7,9,11,13,15,17-octadecanonaene-1,18-diyl)bis | HMDB | | (all-e)-1,1'-(3,7,12,16-Tetramethyl-1,3,5,7,9,11,13,15,17-octadecanonaene-1,18-diyl)bis[2,6,6-trimethyl-cyclohexene | HMDB | | all-e-b-Carotene | HMDB | | all-epsilon-beta-Carotene | HMDB | | Betacarotene | HMDB | | BetaVit | HMDB | | Carotaben | HMDB | | Carotene base 80S | HMDB | | FOOD Orange 5 | HMDB | | KPMK | HMDB | | Lucaratin | HMDB | | Lucarotin | HMDB | | Lurotin | HMDB | | Provatene | HMDB | | Provatenol | HMDB | | Rovimix b-carotene | HMDB | | Serlabo | HMDB | | Solatene | HMDB | | Hermal brand OF betacarotene | HMDB | | Max-caro | HMDB | | Merck brand OF betacarotene | HMDB | | Roche brand OF betacarotene | HMDB | | BellaCarotin | HMDB | | Betacarotene roche brand | HMDB | | Betacarotene solgar brand | HMDB | | Marlyn brand OF betacarotene | HMDB | | Betacarotene hermal brand | HMDB | | Max caro | HMDB | | Solgar brand OF betacarotene | HMDB | | 3m Brand OF betacarotene | HMDB | | Betacarotene 3m brand | HMDB | | Betacarotene marlyn brand | HMDB | | Betacarotene merck brand | HMDB | | Carotene, beta | HMDB | | MaxCaro | HMDB | | Vetoron | HMDB | | beta Carotene | HMDB | | b Carotene | HMDB | | Β carotene | HMDB | | beta-Carotene | HMDB |
|
|---|
| Chemical Formula | C40H56 |
|---|
| Average Molecular Weight | 536.888 |
|---|
| Monoisotopic Molecular Weight | 536.438201803 |
|---|
| IUPAC Name | 1,3,3-trimethyl-2-[(1E,3E,5E,7E,9E,11E,13E,15E,17E)-3,7,12,16-tetramethyl-18-(2,6,6-trimethylcyclohex-1-en-1-yl)octadeca-1,3,5,7,9,11,13,15,17-nonaen-1-yl]cyclohex-1-ene |
|---|
| Traditional Name | β-carotene |
|---|
| CAS Registry Number | 7235-40-7 |
|---|
| SMILES | C\C(\C=C\C=C(/C)\C=C\C1=C(C)CCCC1(C)C)=C/C=C/C=C(\C)/C=C/C=C(\C)/C=C/C1=C(C)CCCC1(C)C |
|---|
| InChI Identifier | InChI=1S/C40H56/c1-31(19-13-21-33(3)25-27-37-35(5)23-15-29-39(37,7)8)17-11-12-18-32(2)20-14-22-34(4)26-28-38-36(6)24-16-30-40(38,9)10/h11-14,17-22,25-28H,15-16,23-24,29-30H2,1-10H3/b12-11+,19-13+,20-14+,27-25+,28-26+,31-17+,32-18+,33-21+,34-22+ |
|---|
| InChI Key | OENHQHLEOONYIE-JLTXGRSLSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | Belongs to the class of organic compounds known as carotenes. These are a type of unsaturated hydrocarbons containing eight consecutive isoprene units. They are characterized by the presence of two end-groups (mostly cyclohexene rings, but also cyclopentene rings or acyclic groups) linked by a long branched alkyl chain. Carotenes belonging form a subgroup of the carotenoids family. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Lipids and lipid-like molecules |
|---|
| Class | Prenol lipids |
|---|
| Sub Class | Tetraterpenoids |
|---|
| Direct Parent | Carotenes |
|---|
| Alternative Parents | |
|---|
| Substituents | - Carotene
- Branched unsaturated hydrocarbon
- Cycloalkene
- Cyclic olefin
- Unsaturated aliphatic hydrocarbon
- Unsaturated hydrocarbon
- Olefin
- Hydrocarbon
- Aliphatic homomonocyclic compound
|
|---|
| Molecular Framework | Aliphatic homomonocyclic compounds |
|---|
| External Descriptors | |
|---|
| Ontology |
|---|
| Physiological effect | Not Available |
|---|
| Disposition | Biological locationSourceExogenousFoodAnimal originVegetableFruitHerb and spiceNutCereal and cereal productPulseGourdSoyTeaBeverageAquatic originEggConfectioneryMilk and milk productOther milk productFermented milkFermented milk productUnfermented milk- Milk (Other mammals) (FooDB: FOOD00690)
- Milk (Human) (FooDB: FOOD00666)
- Milk (Cow) (FooDB: FOOD00618)
- Cow milk, pasteurized, vitamin A + D added, 0% fat (FooDB: FOOD00889)
- Cow milk, pasteurized, vitamin A + D added, 1% fat (FooDB: FOOD00890)
- Cow milk, pasteurized, vitamin A + D added, 2% fat (FooDB: FOOD00891)
- Cow milk, pasteurized, vitamin D added, 3.25% fat (FooDB: FOOD00892)
Fat and oilCocoa and cocoa productBaking goodBaby foodUnclassified food or beverageDishCoffee and coffee productSnack |
|---|
| Process | |
|---|
| Role | |
|---|
| Physical Properties |
|---|
| State | Solid |
|---|
| Experimental Molecular Properties | |
|---|
| Experimental Chromatographic Properties | Not Available |
|---|
| Predicted Molecular Properties | |
|---|
| Predicted Chromatographic Properties | Predicted Collision Cross SectionsPredicted Kovats Retention IndicesUnderivatized |
|---|
| Spectra |
|---|
| GC-MS Spectra| Spectrum Type | Description | Splash Key | Deposition Date | Source | View |
|---|
| Predicted GC-MS | Predicted GC-MS Spectrum - beta-Carotene GC-MS (Non-derivatized) - 70eV, Positive | splash10-00di-3211290000-f968c686d39ae0f0fc50 | 2017-09-01 | Wishart Lab | View Spectrum | | MS | Mass Spectrum (Electron Ionization) | splash10-05mx-9810010000-9a261fb8602890c2481e | 2015-03-01 | Not Available | View Spectrum |
MS/MS Spectra| Spectrum Type | Description | Splash Key | Deposition Date | Source | View |
|---|
| Experimental LC-MS/MS | LC-MS/MS Spectrum - beta-Carotene Quattro_QQQ 10V, Positive-QTOF (Annotated) | splash10-0016-2794670000-c5bc2a4dbcd0f12cbf7f | 2012-07-24 | HMDB team, MONA | View Spectrum | | Experimental LC-MS/MS | LC-MS/MS Spectrum - beta-Carotene Quattro_QQQ 25V, Positive-QTOF (Annotated) | splash10-0002-2687970000-782d048d873c21fc6b82 | 2012-07-24 | HMDB team, MONA | View Spectrum | | Experimental LC-MS/MS | LC-MS/MS Spectrum - beta-Carotene Quattro_QQQ 40V, Positive-QTOF (Annotated) | splash10-05ai-1009510000-585581456b10671692bd | 2012-07-24 | HMDB team, MONA | View Spectrum | | Experimental LC-MS/MS | LC-MS/MS Spectrum - beta-Carotene FAB-EBEB (JMS-HX/HX 110A, JEOL) , Positive-QTOF | splash10-05mx-2920010000-471219f3e66bafb29bee | 2012-08-31 | HMDB team, MONA | View Spectrum | | Experimental LC-MS/MS | LC-MS/MS Spectrum - beta-Carotene , positive-QTOF | splash10-0006-0144900000-2fa7b609c7300ff8aba3 | 2017-09-14 | HMDB team, MONA | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - beta-Carotene 10V, Positive-QTOF | splash10-000i-0434190000-0f2f0f2c4af76e6c3322 | 2016-09-12 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - beta-Carotene 20V, Positive-QTOF | splash10-000b-0679100000-6f4450c8ade1963f9201 | 2016-09-12 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - beta-Carotene 40V, Positive-QTOF | splash10-000j-1789100000-6f2a7024e42303e7737a | 2016-09-12 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - beta-Carotene 10V, Negative-QTOF | splash10-000i-0000090000-cdea7c2d939f0c68b905 | 2016-09-12 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - beta-Carotene 20V, Negative-QTOF | splash10-000i-0000090000-530c81799c9daabdc9bf | 2016-09-12 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - beta-Carotene 40V, Negative-QTOF | splash10-014i-0968170000-ec71ee19e546088e3b05 | 2016-09-12 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - beta-Carotene 10V, Positive-QTOF | splash10-000j-0277960000-5b1eef16ffbc2ccd4b1e | 2021-09-24 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - beta-Carotene 20V, Positive-QTOF | splash10-0081-0449400000-e362d306c4b151321fb6 | 2021-09-24 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - beta-Carotene 40V, Positive-QTOF | splash10-0iki-0149200000-5b858e5700ff1d750a4e | 2021-09-24 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - beta-Carotene 10V, Negative-QTOF | splash10-000i-0001090000-b0a4df7c3417e82842d7 | 2021-09-24 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - beta-Carotene 20V, Negative-QTOF | splash10-000j-0135090000-fb91b96b3605a056f12f | 2021-09-24 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - beta-Carotene 40V, Negative-QTOF | splash10-0apj-0229000000-23e454829ef21fbedf66 | 2021-09-24 | Wishart Lab | View Spectrum |
NMR Spectra| Spectrum Type | Description | Deposition Date | Source | View |
|---|
| Predicted 1D NMR | 13C NMR Spectrum (1D, 100 MHz, H2O, predicted) | 2022-08-20 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 1H NMR Spectrum (1D, 100 MHz, H2O, predicted) | 2022-08-20 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 13C NMR Spectrum (1D, 1000 MHz, H2O, predicted) | 2022-08-20 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 1H NMR Spectrum (1D, 1000 MHz, H2O, predicted) | 2022-08-20 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 13C NMR Spectrum (1D, 200 MHz, H2O, predicted) | 2022-08-20 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 1H NMR Spectrum (1D, 200 MHz, H2O, predicted) | 2022-08-20 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 13C NMR Spectrum (1D, 300 MHz, H2O, predicted) | 2022-08-20 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 1H NMR Spectrum (1D, 300 MHz, H2O, predicted) | 2022-08-20 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 13C NMR Spectrum (1D, 400 MHz, H2O, predicted) | 2022-08-20 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 1H NMR Spectrum (1D, 400 MHz, H2O, predicted) | 2022-08-20 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 13C NMR Spectrum (1D, 500 MHz, H2O, predicted) | 2022-08-20 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 1H NMR Spectrum (1D, 500 MHz, H2O, predicted) | 2022-08-20 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 13C NMR Spectrum (1D, 600 MHz, H2O, predicted) | 2022-08-20 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 1H NMR Spectrum (1D, 600 MHz, H2O, predicted) | 2022-08-20 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 13C NMR Spectrum (1D, 700 MHz, H2O, predicted) | 2022-08-20 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 1H NMR Spectrum (1D, 700 MHz, H2O, predicted) | 2022-08-20 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 13C NMR Spectrum (1D, 800 MHz, H2O, predicted) | 2022-08-20 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 1H NMR Spectrum (1D, 800 MHz, H2O, predicted) | 2022-08-20 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 13C NMR Spectrum (1D, 900 MHz, H2O, predicted) | 2022-08-20 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 1H NMR Spectrum (1D, 900 MHz, H2O, predicted) | 2022-08-20 | Wishart Lab | View Spectrum | | Experimental 2D NMR | [1H, 13C]-HSQC NMR Spectrum (2D, 600 MHz, CDCl3, experimental) | 2012-12-05 | Wishart Lab | View Spectrum |
|
|---|
| Biological Properties |
|---|
| Cellular Locations | - Cytoplasm
- Extracellular
- Membrane (predicted from logP)
|
|---|
| Biospecimen Locations | |
|---|
| Tissue Locations | - Adipose Tissue
- Adrenal Gland
- Epidermis
- Erythrocyte
- Fibroblasts
- Intestine
- Liver
- Placenta
- Platelet
- Prostate
- Spleen
|
|---|
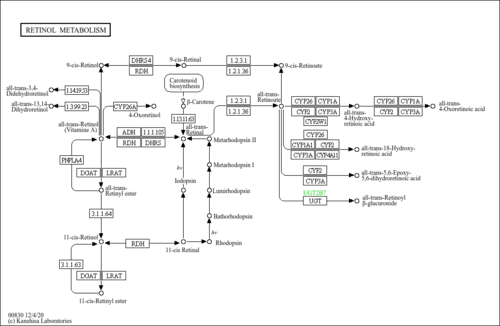
| Pathways | |
|---|
| Normal Concentrations |
|---|
| |
| Blood | Detected and Quantified | 0.133 uM | Adult (>18 years old) | Both | Normal | | details | | Blood | Detected and Quantified | 0.936 +/- 0.0300 uM | Adult (>18 years old) | Female | Normal | | details | | Blood | Detected and Quantified | 0.420 +/- 0.330 uM | Adult (>18 years old) | Both | Normal | | details | | Blood | Detected and Quantified | 0.450 +/- 0.250 uM | Adult (>18 years old) | Both | Normal | | details | | Blood | Detected and Quantified | 0.460 +/- 0.220 uM | Adult (>18 years old) | Both | Normal | | details | | Blood | Detected and Quantified | 0.480 +/- 0.250 uM | Adult (>18 years old) | Both | Normal | | details | | Blood | Detected and Quantified | 0.304 +/- 0.340 uM | Adult (>18 years old) | Female | Normal | | details | | Blood | Detected and Quantified | 0.400 +/- 0.210 uM | Adult (>18 years old) | Female | Normal | | details | | Blood | Detected and Quantified | 0.510 +/- 0.340 uM | Adult (>18 years old) | Female | Normal | | details | | Blood | Detected and Quantified | 0.196 +/- 0.230 uM | Adult (>18 years old) | Male | Normal | | details | | Blood | Detected and Quantified | 0.330 +/- 0.230 uM | Adult (>18 years old) | Male | Normal | | details | | Blood | Detected and Quantified | 0.510 +/- 0.380 uM | Adult (>18 years old) | Male | Normal | | details | | Blood | Detected and Quantified | 0.520 +/- 0.480 uM | Adult (>18 years old) | Male | Normal | | details | | Blood | Detected and Quantified | 0.82 +/- 0.58 uM | Children (1-13 years old) | Both | Normal | | details | | Blood | Detected and Quantified | 0.393 +/- 0.530 uM | Adult (>18 years old) | Not Specified | Normal | | details | | Blood | Detected and Quantified | 1.59 +/- 0.53 uM | Adult (>18 years old) | Male | Normal | | details | | Blood | Detected and Quantified | 0.653 +/- 0.355 uM | Adult (>18 years old) | Female | Normal | | details | | Blood | Detected and Quantified | 1.75 +/- 0.64 uM | Adult (>18 years old) | Female | Normal | | details | | Blood | Detected and Quantified | 0.391 +/- 0.215 uM | Adult (>18 years old) | Female | Normal | | details | | Blood | Detected and Quantified | 0.685 +/- 0.404 uM | Adult (>18 years old) | Female | Normal | | details | | Blood | Detected and Quantified | 0.440 +/- 0.251 uM | Adult (>18 years old) | Female | Normal | | details | | Blood | Detected and Quantified | 0.560 +/- 0.440 uM | Adult (>18 years old) | Female | Normal | | details | | Blood | Detected and Quantified | 0.410 +/- 0.340 uM | Adult (>18 years old) | Male | Normal | | details | | Blood | Detected and Quantified | 0.460 +/- 0.304 uM | Adult (>18 years old) | Both | Normal | | details | | Blood | Detected and Quantified | 0.580 +/- 0.480 uM | Adult (>18 years old) | Female | Normal | | details | | Blood | Detected and Quantified | 0.640 +/- 0.440 uM | Adult (>18 years old) | Female | Normal | | details | | Blood | Detected and Quantified | 0.26 uM | Adult (>18 years old) | Female | Normal | | details | | Blood | Detected and Quantified | 0.21 uM | Adult (>18 years old) | Male | Normal | | details | | Blood | Detected and Quantified | 340 +/- 190 uM | Adult (>18 years old) | Female | Normal | | details | | Blood | Detected and Quantified | 0.421 +/- 0.253 uM | Adult (>18 years old) | Both | Normal | | details | | Blood | Detected and Quantified | 0.260 +/- 0.120 uM | Adult (>18 years old) | Female | Normal | | details | | Blood | Detected and Quantified | 0.230 +/- 0.120 uM | Adult (>18 years old) | Male | Normal | | details | | Blood | Detected and Quantified | 0.213 +/- 0.190 uM | Adult (>18 years old) | Both | Normal | | details | | Blood | Detected and Quantified | 0.31 uM | Adult (>18 years old) | Both | Normal | | details | | Blood | Detected and Quantified | 0.33 uM | Adult (>18 years old) | Both | Normal | | details | | Blood | Detected and Quantified | 0.19 uM | Adult (>18 years old) | Both | Normal | | details | | Blood | Detected and Quantified | 0.748 uM | Adult (>18 years old) | Female | Normal | | details | | Blood | Detected and Quantified | 1.391 uM | Adult (>18 years old) | Female | Normal | | details | | Blood | Detected and Quantified | 1.4 uM | Adult (>18 years old) | Female | Normal | | details | | Blood | Detected and Quantified | 0.844 uM | Adult (>18 years old) | Female | Normal | | details | | Blood | Detected and Quantified | 0.943 uM | Adult (>18 years old) | Female | Normal | | details | | Blood | Detected and Quantified | 1.071 uM | Adult (>18 years old) | Female | Normal | | details | | Blood | Detected and Quantified | 0.56 (0.23-3.92) uM | Adolescent (13-18 years old) | Both | Normal | | details | | Blood | Detected and Quantified | 0.414 uM | Adult (>18 years old) | Both | Normal | | details | | Blood | Detected and Quantified | 0.501 uM | Adult (>18 years old) | Both | Normal | | details | | Blood | Detected and Quantified | 0.528 uM | Adult (>18 years old) | Both | Normal | | details | | Blood | Detected and Quantified | 0.83 uM | Adult (>18 years old) | Both | Normal | | details | | Blood | Detected and Quantified | 0.84 uM | Adult (>18 years old) | Both | Normal | | details | | Blood | Detected and Quantified | 0.85 uM | Adult (>18 years old) | Both | Normal | | details | | Blood | Detected and Quantified | 0.89 uM | Adult (>18 years old) | Both | Normal | | details | | Blood | Detected and Quantified | 0.198 +/- 0.150 uM | Adult (>18 years old) | Both | Normal | | details | | Blood | Detected and Quantified | 0.292 +/- 0.218 uM | Adult (>18 years old) | Both | Normal | | details | | Blood | Detected and Quantified | 0.370 +/- 0.290 uM | Adult (>18 years old) | Both | Normal | | details | | Blood | Detected and Quantified | 0.400 +/- 0.340 uM | Adult (>18 years old) | Both | Normal | | details | | Blood | Detected and Quantified | 0.410 +/- 0.270 uM | Adult (>18 years old) | Both | Normal | | details | | Blood | Detected and Quantified | 0.420 +/- 0.340 uM | Adult (>18 years old) | Both | Normal | | details | | Blood | Detected and Quantified | 0.430 +/- 0.360 uM | Adult (>18 years old) | Both | Normal | | details | | Blood | Detected and Quantified | 0.440 +/- 0.471 uM | Adult (>18 years old) | Both | Normal | | details | | Blood | Detected and Quantified | 0.450 +/- 0.290 uM | Adult (>18 years old) | Both | Normal | | details | | Blood | Detected and Quantified | 0.451 +/- 0.292 uM | Adult (>18 years old) | Both | Normal | | details | | Blood | Detected and Quantified | 0.490 +/- 0.380 uM | Adult (>18 years old) | Both | Normal | | details | | Blood | Detected and Quantified | 0.530 +/- 0.350 uM | Adult (>18 years old) | Both | Normal | | details | | Blood | Detected and Quantified | 0.540 +/- 0.340 uM | Adult (>18 years old) | Both | Normal | | details | | Blood | Detected and Quantified | 0.620 +/- 0.590 uM | Adult (>18 years old) | Both | Normal | | details | | Blood | Detected and Quantified | 0.728 +/- 0.358 uM | Adult (>18 years old) | Both | Normal | | details | | Blood | Detected and Quantified | 0.164 +/- 0.120 uM | Adult (>18 years old) | Both | Normal | | details | | Blood | Detected and Quantified | 0.333 uM | Adolescent (13-18 years old) | Female | Normal | | details | | Blood | Detected and Quantified | 0.374 uM | Children (1-13 years old) | Female | Normal | | details | | Blood | Detected and Quantified | 0.44 uM | Adult (>18 years old) | Female | Normal | | details | | Blood | Detected and Quantified | 0.536 uM | Adult (>18 years old) | Female | Normal | | details | | Blood | Detected and Quantified | 0.54 uM | Adult (>18 years old) | Female | Normal | | details | | Blood | Detected and Quantified | 0.71 uM | Adult (>18 years old) | Female | Normal | | details | | Blood | Detected and Quantified | 0.72 uM | Adult (>18 years old) | Female | Normal | | details | | Blood | Detected and Quantified | 0.302 +/- 0.184 uM | Adult (>18 years old) | Female | Normal | | details | | Blood | Detected and Quantified | 0.373 +/- 0.319 uM | Adult (>18 years old) | Female | Normal | | details | | Blood | Detected and Quantified | 0.397 +/- 0.384 uM | Adult (>18 years old) | Female | Normal | | details | | Blood | Detected and Quantified | 0.406 +/- 0.345 uM | Adult (>18 years old) | Female | Normal | | details | | Blood | Detected and Quantified | 0.471 +/- 0.309 uM | Adult (>18 years old) | Female | Normal | | details | | Blood | Detected and Quantified | 0.542 +/- 0.270 uM | Adult (>18 years old) | Female | Normal | | details | | Blood | Detected and Quantified | 0.580 +/- 0.370 uM | Adult (>18 years old) | Female | Normal | | details | | Blood | Detected and Quantified | 0.581 +/- 0.311 uM | Adult (>18 years old) | Female | Normal | | details | | Blood | Detected and Quantified | 0.600 +/- 0.500 uM | Adolescent (13-18 years old) | Female | Normal | | details | | Blood | Detected and Quantified | 0.821 +/- 0.725 uM | Adult (>18 years old) | Female | Normal | | details | | Blood | Detected and Quantified | 574.980 +/- 375.125 uM | Adult (>18 years old) | Female | Normal | | details | | Blood | Detected and Quantified | 1061.674 +/- 689.157 uM | Adult (>18 years old) | Female | Normal | | details | | Blood | Detected and Quantified | 0.307 uM | Adolescent (13-18 years old) | Male | Normal | | details | | Blood | Detected and Quantified | 0.33 uM | Adult (>18 years old) | Male | Normal | | details | | Blood | Detected and Quantified | 0.36 uM | Adult (>18 years old) | Male | Normal | | details | | Blood | Detected and Quantified | 0.434 uM | Children (1-13 years old) | Male | Normal | | details | | Blood | Detected and Quantified | 0.48 uM | Adult (>18 years old) | Male | Normal | | details | | Blood | Detected and Quantified | 0.62 uM | Adult (>18 years old) | Male | Normal | | details | | Blood | Detected and Quantified | 0.209 +/- 0.184 uM | Adult (>18 years old) | Male | Normal | | details | | Blood | Detected and Quantified | 0.240 +/- 0.152 uM | Adult (>18 years old) | Male | Normal | | details | | Blood | Detected and Quantified | 0.244 +/- 0.207 uM | Adult (>18 years old) | Male | Normal | | details | | Blood | Detected and Quantified | 0.251 +/- 0.212 uM | Adult (>18 years old) | Male | Normal | | details | | Blood | Detected and Quantified | 0.257 +/- 0.235 uM | Adult (>18 years old) | Male | Normal | | details | | Blood | Detected and Quantified | 0.283 +/- 0.143 uM | Adult (>18 years old) | Male | Normal | | details | | Blood | Detected and Quantified | 0.357 +/- 0.285 uM | Adult (>18 years old) | Male | Normal | | details | | Blood | Detected and Quantified | 0.449 +/- 0.230 uM | Adult (>18 years old) | Male | Normal | | details | | Blood | Detected and Quantified | 0.460 +/- 0.290 uM | Adult (>18 years old) | Male | Normal | | details | | Blood | Detected and Quantified | 0.470 +/- 0.330 uM | Adolescent (13-18 years old) | Male | Normal | | details | | Blood | Detected and Quantified | 0.484 +/- 0.407 uM | Adult (>18 years old) | Male | Normal | | details | | Blood | Detected and Quantified | 350 +/- 150 uM | Adult (>18 years old) | Male | Normal | | details | | Blood | Detected and Quantified | 459.500 +/- 290.191 uM | Adult (>18 years old) | Male | Normal | | details | | Blood | Detected and Quantified | 580 +/- 300 uM | Adult (>18 years old) | Male | Normal | | details | | Blood | Detected and Quantified | 596.0275 +/- 447.0206 uM | Adult (>18 years old) | Male | Normal | | details | | Blood | Detected and Quantified | 0.590 +/- 0.440 uM | Children (1 - 13 years old) | Not Specified | Normal | | details | | Blood | Detected and Quantified | 0.03-2.70 uM | Adult (>18 years old) | Both | Normal | | details | | Blood | Detected and Quantified | 0.42 (0.19-0.72) uM | Adult (>18 years old) | Female | Normal | | details | | Blood | Detected and Quantified | 1.18 +/- 0.06 uM | Adult (>18 years old) | Both | Normal | | details | | Blood | Detected and Quantified | 1.01 +/- 0.08 uM | Adult (>18 years old) | Male | Normal | | details | | Blood | Detected and Quantified | 1.0 (0.32 - 1.83) uM | Adult (>18 years old) | Not Specified | Normal | | details | | Blood | Detected and Quantified | 0.23 uM | Adult (>18 years old) | Both | Normal | | details | | Blood | Detected and Quantified | 0.28 uM | Adult (>18 years old) | Both | Normal | | details | | Breast Milk | Detected but not Quantified | Not Quantified | Adult (>18 years old) | Female | Normal | | details | | Feces | Detected but not Quantified | Not Quantified | Adult (>18 years old) | Both | Normal | | details |
|
|---|
| Abnormal Concentrations |
|---|
| |
| Blood | Detected and Quantified | 0.201 +/- 0.180 uM | Adult (>18 years old) | Not Specified | Obese | | details | | Blood | Detected and Quantified | 0.249 +/- 0.250 uM | Adult (>18 years old) | Not Specified | Overweight | | details | | Blood | Detected and Quantified | 277000 uM | Adult (>18 years old) | Both | Colorectal, breast, or neuroendocrine cancer | | details | | Blood | Detected and Quantified | 0.390 +/- 0.260 uM | Adult (>18 years old) | Both | Type I diabetes | | details | | Blood | Detected and Quantified | 0.200 +/- 0.130 uM | Children (1 - 13 years old) | Not Specified | Obese | | details | | Blood | Detected and Quantified | 0.330 +/- 0.190 uM | Children (1 - 13 years old) | Not Specified | Overweight | | details | | Blood | Detected and Quantified | 0.30 (0.15-0.45) uM | Adult (>18 years old) | Female | Endometrial cancer | | details |
|
|---|
| Associated Disorders and Diseases |
|---|
| Disease References | | Endometrial cancer |
|---|
- Jeong NH, Song ES, Lee JM, Lee KB, Kim MK, Yun YM, Lee JK, Son SK, Lee JP, Kim JH, Hur SY, Kwon YI: Preoperative levels of plasma micronutrients are related to endometrial cancer risk. Acta Obstet Gynecol Scand. 2009;88(4):434-9. doi: 10.1080/00016340902767187. [PubMed:19235557 ]
| | Obesity |
|---|
- Vioque J, Weinbrenner T, Asensio L, Castello A, Young IS, Fletcher A: Plasma concentrations of carotenoids and vitamin C are better correlated with dietary intake in normal weight than overweight and obese elderly subjects. Br J Nutr. 2007 May;97(5):977-86. [PubMed:17408529 ]
- Burrows TL, Warren JM, Colyvas K, Garg ML, Collins CE: Validation of overweight children's fruit and vegetable intake using plasma carotenoids. Obesity (Silver Spring). 2009 Jan;17(1):162-8. doi: 10.1038/oby.2008.495. Epub 2008 Nov 6. [PubMed:18997681 ]
| | Diabetes mellitus type 1 |
|---|
- Granado-Lorencio F, Olmedilla-Alonso B, Blanco-Navarro I, Botella-Romero F, Simal-Anton A: Assessment of carotenoid status and the relation to glycaemic control in type I diabetics: a follow-up study. Eur J Clin Nutr. 2006 Aug;60(8):1000-8. Epub 2006 Feb 1. [PubMed:16452910 ]
|
|
|---|
| Associated OMIM IDs | |
|---|
| External Links |
|---|
| DrugBank ID | DB06755 |
|---|
| Phenol Explorer Compound ID | Not Available |
|---|
| FooDB ID | FDB014613 |
|---|
| KNApSAcK ID | C00000919 |
|---|
| Chemspider ID | 4444129 |
|---|
| KEGG Compound ID | C02094 |
|---|
| BioCyc ID | CPD1F-129 |
|---|
| BiGG ID | Not Available |
|---|
| Wikipedia Link | Beta_Carotene |
|---|
| METLIN ID | Not Available |
|---|
| PubChem Compound | 5280489 |
|---|
| PDB ID | Not Available |
|---|
| ChEBI ID | 17579 |
|---|
| Food Biomarker Ontology | Not Available |
|---|
| VMH ID | CARO |
|---|
| MarkerDB ID | MDB00029986 |
|---|
| Good Scents ID | rw1105911 |
|---|
| References |
|---|
| Synthesis Reference | Not Available |
|---|
| Material Safety Data Sheet (MSDS) | Download (PDF) |
|---|
| General References | - Rushin WG, Catignani GL, Schwartz SJ: Determination of beta-carotene and its cis isomers in serum. Clin Chem. 1990 Nov;36(11):1986-9. [PubMed:2242585 ]
- Vahlquist A, Lee JB, Michaelsson G, Rollman O: Vitamin A in human skin: II Concentrations of carotene, retinol and dehydroretinol in various components of normal skin. J Invest Dermatol. 1982 Aug;79(2):94-7. [PubMed:7097042 ]
- Bacic-Vrca V, Skreb F, Cepelak I, Mayer L, Kusic Z, Petres B: The effect of antioxidant supplementation on superoxide dismutase activity, Cu and Zn levels, and total antioxidant status in erythrocytes of patients with Graves' disease. Clin Chem Lab Med. 2005;43(4):383-8. [PubMed:15899653 ]
- Wu K, Erdman JW Jr, Schwartz SJ, Platz EA, Leitzmann M, Clinton SK, DeGroff V, Willett WC, Giovannucci E: Plasma and dietary carotenoids, and the risk of prostate cancer: a nested case-control study. Cancer Epidemiol Biomarkers Prev. 2004 Feb;13(2):260-9. [PubMed:14973107 ]
- Huang C, Tang YL, Chen CY, Chen ML, Chu CH, Hseu CT: The bioavailability of beta-carotene in stir- or deep-fried vegetables in men determined by measuring the serum response to a single ingestion. J Nutr. 2000 Mar;130(3):534-40. [PubMed:10702581 ]
- Gerber M: Qualitative methods to evaluate Mediterranean diet in adults. Public Health Nutr. 2006 Feb;9(1A):147-51. [PubMed:16512962 ]
- El-Sohemy A, Baylin A, Kabagambe E, Ascherio A, Spiegelman D, Campos H: Individual carotenoid concentrations in adipose tissue and plasma as biomarkers of dietary intake. Am J Clin Nutr. 2002 Jul;76(1):172-9. [PubMed:12081831 ]
- Gollnick HP, Siebenwirth C: Beta-carotene plasma levels and content in oral mucosal epithelium is skin type associated. Skin Pharmacol Appl Skin Physiol. 2002 Sep-Oct;15(5):360-6. [PubMed:12239432 ]
- Lee J, Jiang S, Levine N, Watson RR: Carotenoid supplementation reduces erythema in human skin after simulated solar radiation exposure. Proc Soc Exp Biol Med. 2000 Feb;223(2):170-4. [PubMed:10654620 ]
- Stahl W, Schwarz W, Sundquist AR, Sies H: cis-trans isomers of lycopene and beta-carotene in human serum and tissues. Arch Biochem Biophys. 1992 Apr;294(1):173-7. [PubMed:1550343 ]
- Stahl W, Heinrich U, Jungmann H, Sies H, Tronnier H: Carotenoids and carotenoids plus vitamin E protect against ultraviolet light-induced erythema in humans. Am J Clin Nutr. 2000 Mar;71(3):795-8. [PubMed:10702175 ]
- Chanarat N, Chanarat P, Viratsethasin K, Suttajit M, Chiewsilp D: Biochemical and hematological manifestations of HIV/AIDS in Chiang Mai, Thailand. Southeast Asian J Trop Med Public Health. 2001 Sep;32(3):500-3. [PubMed:11944706 ]
- Heinrich U, Gartner C, Wiebusch M, Eichler O, Sies H, Tronnier H, Stahl W: Supplementation with beta-carotene or a similar amount of mixed carotenoids protects humans from UV-induced erythema. J Nutr. 2003 Jan;133(1):98-101. [PubMed:12514275 ]
- Poor CL, Bierer TL, Merchen NR, Fahey GC Jr, Murphy MR, Erdman JW Jr: Evaluation of the preruminant calf as a model for the study of human carotenoid metabolism. J Nutr. 1992 Feb;122(2):262-8. [PubMed:1732467 ]
- Lakshman MR: Alpha and omega of carotenoid cleavage. J Nutr. 2004 Jan;134(1):241S-245S. [PubMed:14704327 ]
- Kirsh VA, Hayes RB, Mayne ST, Chatterjee N, Subar AF, Dixon LB, Albanes D, Andriole GL, Urban DA, Peters U: Supplemental and dietary vitamin E, beta-carotene, and vitamin C intakes and prostate cancer risk. J Natl Cancer Inst. 2006 Feb 15;98(4):245-54. [PubMed:16478743 ]
- McArdle F, Rhodes LE, Parslew RA, Close GL, Jack CI, Friedmann PS, Jackson MJ: Effects of oral vitamin E and beta-carotene supplementation on ultraviolet radiation-induced oxidative stress in human skin. Am J Clin Nutr. 2004 Nov;80(5):1270-5. [PubMed:15531675 ]
- Metnitz GH, Fischer M, Bartens C, Steltzer H, Lang T, Druml W: Impact of acute renal failure on antioxidant status in multiple organ failure. Acta Anaesthesiol Scand. 2000 Mar;44(3):236-40. [PubMed:10714834 ]
- Zhou JF, Chen P, Yang JL, Zhu YG, Peng CH, Wu YL: Oxidative stress before and after operation in patients with chronic cholecystitis containing gallstone. Biomed Environ Sci. 2000 Dec;13(4):254-62. [PubMed:11351858 ]
- Dimenstein R, Trugo NM, Donangelo CM, Trugo LC, Anastacio AS: Effect of subadequate maternal vitamin-A status on placental transfer of retinol and beta-carotene to the human fetus. Biol Neonate. 1996;69(4):230-4. [PubMed:8724650 ]
|
|---|