| Record Information |
|---|
| Version | 5.0 |
|---|
| Status | Expected but not Quantified |
|---|
| Creation Date | 2006-08-13 07:49:22 UTC |
|---|
| Update Date | 2022-03-07 02:49:20 UTC |
|---|
| HMDB ID | HMDB0003941 |
|---|
| Secondary Accession Numbers | |
|---|
| Metabolite Identification |
|---|
| Common Name | 3-Oxooctanoyl-CoA |
|---|
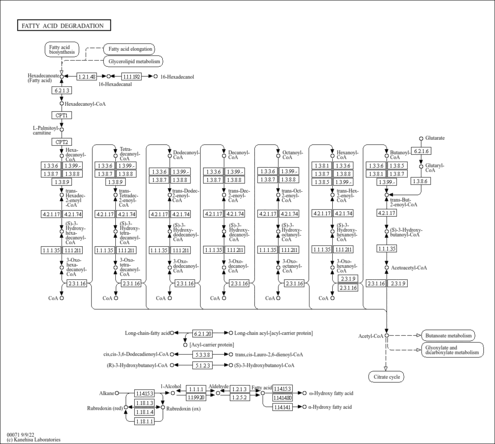
| Description | 3-Oxooctanoyl-CoA is the substrate of the acetyl-CoA C-acyltransferase/oxoacyl-CoA thiolase A (EC 2.3.1.16, SCP2/3-oxoacyl-CoA thiolase) present in peroxisomes from normal liver. Peroxisomes beta -oxidize a wide variety of substrates including straight chain fatty acids, 2-methyl-branched fatty acids, and the side chain of the bile acid intermediates di- and trihydroxycoprostanic acids. Peroxisomes contain several beta -oxidation pathways with different substrate specificities; or example, straight chain acyl-CoAs are desaturated by palmitoyl-CoA oxidase, and their enoyl-CoAs are then converted to 3-oxoacyl-CoAs by MFP-1, which forms (hydration) and dehydrogenates L-3(3S)-hydroxyacyl-CoAs; for example, straight chain acyl-CoAs are desaturated by palmitoyl-CoA oxidase (23), and their enoyl-CoAs are then converted to 3-oxoacyl-CoAs by 3-hydroxyacyl-CoA dehydrogenase (EC 1.1.1.35), which forms (hydration) and dehydrogenates L-3(3S)-hydroxyacyl-CoAs and their enoyl-CoAs are then converted to the corresponding 3-oxoacyl-CoAs by long-chain-enoyl-CoA hydratase(EC 4.2.1.74), which forms and dehydrogenates D-3(3R)-hydroxyacyl-CoAs. (PMID: 9325339 ). |
|---|
| Structure | CCCCCC(=O)CC(=O)SCCNC(=O)CCNC(=O)C(O)C(C)(C)COP(O)(=O)OP(O)(=O)OC[C@H]1O[C@H]([C@H](O)[C@@H]1OP(O)(O)=O)N1C=NC2=C(N)N=CN=C12 InChI=1S/C29H48N7O18P3S/c1-4-5-6-7-17(37)12-20(39)58-11-10-31-19(38)8-9-32-27(42)24(41)29(2,3)14-51-57(48,49)54-56(46,47)50-13-18-23(53-55(43,44)45)22(40)28(52-18)36-16-35-21-25(30)33-15-34-26(21)36/h15-16,18,22-24,28,40-41H,4-14H2,1-3H3,(H,31,38)(H,32,42)(H,46,47)(H,48,49)(H2,30,33,34)(H2,43,44,45)/t18-,22-,23-,24?,28-/m1/s1 |
|---|
| Synonyms | | Value | Source |
|---|
| 3-Ketooctanoyl-CoA | HMDB | | 3-Ketooctanoyl-coenzyme A | HMDB | | 3-Oxooctanoyl coenzyme A | HMDB | | 3-Oxooctanoyl-coenzyme A | HMDB | | S-(3-Oxooctanoate | HMDB | | S-(3-Oxooctanoate) CoA | HMDB | | S-(3-Oxooctanoate) coenzyme A | HMDB | | S-(3-Oxooctanoic acid | HMDB |
|
|---|
| Chemical Formula | C29H48N7O18P3S |
|---|
| Average Molecular Weight | 907.714 |
|---|
| Monoisotopic Molecular Weight | 907.198937993 |
|---|
| IUPAC Name | {[(2R,3S,4R,5R)-5-(6-amino-9H-purin-9-yl)-4-hydroxy-2-({[hydroxy({[hydroxy(3-hydroxy-2,2-dimethyl-3-{[2-({2-[(3-oxooctanoyl)sulfanyl]ethyl}carbamoyl)ethyl]carbamoyl}propoxy)phosphoryl]oxy})phosphoryl]oxy}methyl)oxolan-3-yl]oxy}phosphonic acid |
|---|
| Traditional Name | [(2R,3S,4R,5R)-5-(6-aminopurin-9-yl)-4-hydroxy-2-[({hydroxy[hydroxy(3-hydroxy-2,2-dimethyl-3-{[2-({2-[(3-oxooctanoyl)sulfanyl]ethyl}carbamoyl)ethyl]carbamoyl}propoxy)phosphoryl]oxyphosphoryl}oxy)methyl]oxolan-3-yl]oxyphosphonic acid |
|---|
| CAS Registry Number | 54684-64-9 |
|---|
| SMILES | CCCCCC(=O)CC(=O)SCCNC(=O)CCNC(=O)C(O)C(C)(C)COP(O)(=O)OP(O)(=O)OC[C@H]1O[C@H]([C@H](O)[C@@H]1OP(O)(O)=O)N1C=NC2=C(N)N=CN=C12 |
|---|
| InChI Identifier | InChI=1S/C29H48N7O18P3S/c1-4-5-6-7-17(37)12-20(39)58-11-10-31-19(38)8-9-32-27(42)24(41)29(2,3)14-51-57(48,49)54-56(46,47)50-13-18-23(53-55(43,44)45)22(40)28(52-18)36-16-35-21-25(30)33-15-34-26(21)36/h15-16,18,22-24,28,40-41H,4-14H2,1-3H3,(H,31,38)(H,32,42)(H,46,47)(H,48,49)(H2,30,33,34)(H2,43,44,45)/t18-,22-,23-,24?,28-/m1/s1 |
|---|
| InChI Key | WPIVBCGRGVNDDT-FFJUWABQSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | Belongs to the class of organic compounds known as 3-oxo-acyl coas. These are organic compounds containing a 3-oxo acylated coenzyme A derivative. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Lipids and lipid-like molecules |
|---|
| Class | Fatty Acyls |
|---|
| Sub Class | Fatty acyl thioesters |
|---|
| Direct Parent | 3-oxo-acyl CoAs |
|---|
| Alternative Parents | |
|---|
| Substituents | - Coenzyme a or derivatives
- Purine ribonucleoside 3',5'-bisphosphate
- Purine ribonucleoside bisphosphate
- Purine ribonucleoside diphosphate
- Ribonucleoside 3'-phosphate
- Pentose phosphate
- Pentose-5-phosphate
- Beta amino acid or derivatives
- Glycosyl compound
- N-glycosyl compound
- 6-aminopurine
- Monosaccharide phosphate
- Organic pyrophosphate
- Pentose monosaccharide
- Imidazopyrimidine
- Purine
- Monoalkyl phosphate
- Aminopyrimidine
- 1,3-dicarbonyl compound
- Imidolactam
- Monosaccharide
- N-acyl-amine
- N-substituted imidazole
- Organic phosphoric acid derivative
- Pyrimidine
- Alkyl phosphate
- Fatty amide
- Phosphoric acid ester
- Tetrahydrofuran
- Imidazole
- Azole
- Heteroaromatic compound
- Carbothioic s-ester
- Secondary alcohol
- Ketone
- Thiocarboxylic acid ester
- Carboxamide group
- Secondary carboxylic acid amide
- Amino acid or derivatives
- Sulfenyl compound
- Thiocarboxylic acid or derivatives
- Organoheterocyclic compound
- Azacycle
- Oxacycle
- Carboxylic acid derivative
- Organosulfur compound
- Organic oxygen compound
- Hydrocarbon derivative
- Carbonyl group
- Organic nitrogen compound
- Primary amine
- Organopnictogen compound
- Organic oxide
- Organooxygen compound
- Organonitrogen compound
- Amine
- Alcohol
- Aromatic heteropolycyclic compound
|
|---|
| Molecular Framework | Aromatic heteropolycyclic compounds |
|---|
| External Descriptors | |
|---|
| Ontology |
|---|
| Physiological effect | Not Available |
|---|
| Disposition | |
|---|
| Process | |
|---|
| Role | |
|---|
| Physical Properties |
|---|
| State | Solid |
|---|
| Experimental Molecular Properties | | Property | Value | Reference |
|---|
| Melting Point | Not Available | Not Available | | Boiling Point | Not Available | Not Available | | Water Solubility | Not Available | Not Available | | LogP | -2.164 | Not Available |
|
|---|
| Experimental Chromatographic Properties | Not Available |
|---|
| Predicted Molecular Properties | |
|---|
| Predicted Chromatographic Properties | Predicted Collision Cross SectionsPredicted Kovats Retention IndicesNot Available |
|---|
| GC-MS Spectra| Spectrum Type | Description | Splash Key | Deposition Date | Source | View |
|---|
| MS | Mass Spectrum (Electron Ionization) | Not Available | 2022-08-06 | Not Available | View Spectrum |
MS/MS Spectra| Spectrum Type | Description | Splash Key | Deposition Date | Source | View |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 3-Oxooctanoyl-CoA 10V, Positive-QTOF | splash10-000i-1920100120-cd410d4a53d934763ea7 | 2015-09-14 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 3-Oxooctanoyl-CoA 20V, Positive-QTOF | splash10-000i-0941200000-7cb4acb73a370d1909bf | 2015-09-14 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 3-Oxooctanoyl-CoA 40V, Positive-QTOF | splash10-000i-2920000000-eba79c033775684afd01 | 2015-09-14 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 3-Oxooctanoyl-CoA 10V, Negative-QTOF | splash10-055r-2911020222-0c38a22725feacf64342 | 2015-09-15 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 3-Oxooctanoyl-CoA 20V, Negative-QTOF | splash10-001r-2910110010-a4bf55bed0c3fdbf24bb | 2015-09-15 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 3-Oxooctanoyl-CoA 40V, Negative-QTOF | splash10-057i-6900100000-c125ac237096994d43da | 2015-09-15 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 3-Oxooctanoyl-CoA 10V, Positive-QTOF | splash10-0a4l-1000000179-db3b7929eaea483272f5 | 2021-09-22 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 3-Oxooctanoyl-CoA 20V, Positive-QTOF | splash10-01vo-3000100390-9b0ff1cd5679895e1bbb | 2021-09-22 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 3-Oxooctanoyl-CoA 40V, Positive-QTOF | splash10-0udi-0012900000-4c7bfbc65ade18bd6629 | 2021-09-22 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 3-Oxooctanoyl-CoA 10V, Negative-QTOF | splash10-0a4i-0000000019-26cf0c2172326ab3f7df | 2021-09-23 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 3-Oxooctanoyl-CoA 20V, Negative-QTOF | splash10-0a70-9600201564-fe51baa1e537efc0bcb2 | 2021-09-23 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 3-Oxooctanoyl-CoA 40V, Negative-QTOF | splash10-0174-9203401730-42be20d3aaf464dadd7a | 2021-09-23 | Wishart Lab | View Spectrum |
NMR Spectra| Spectrum Type | Description | Deposition Date | Source | View |
|---|
| Predicted 1D NMR | 13C NMR Spectrum (1D, 100 MHz, D2O, predicted) | 2021-09-24 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 1H NMR Spectrum (1D, 100 MHz, D2O, predicted) | 2021-09-24 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 13C NMR Spectrum (1D, 1000 MHz, D2O, predicted) | 2021-09-24 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 1H NMR Spectrum (1D, 1000 MHz, D2O, predicted) | 2021-09-24 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 13C NMR Spectrum (1D, 200 MHz, D2O, predicted) | 2021-09-24 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 1H NMR Spectrum (1D, 200 MHz, D2O, predicted) | 2021-09-24 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 13C NMR Spectrum (1D, 300 MHz, D2O, predicted) | 2021-09-24 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 1H NMR Spectrum (1D, 300 MHz, D2O, predicted) | 2021-09-24 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 13C NMR Spectrum (1D, 400 MHz, D2O, predicted) | 2021-09-24 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 1H NMR Spectrum (1D, 400 MHz, D2O, predicted) | 2021-09-24 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 13C NMR Spectrum (1D, 500 MHz, D2O, predicted) | 2021-09-24 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 1H NMR Spectrum (1D, 500 MHz, D2O, predicted) | 2021-09-24 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 13C NMR Spectrum (1D, 600 MHz, D2O, predicted) | 2021-09-24 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 1H NMR Spectrum (1D, 600 MHz, D2O, predicted) | 2021-09-24 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 13C NMR Spectrum (1D, 700 MHz, D2O, predicted) | 2021-09-24 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 1H NMR Spectrum (1D, 700 MHz, D2O, predicted) | 2021-09-24 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 13C NMR Spectrum (1D, 800 MHz, D2O, predicted) | 2021-09-24 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 1H NMR Spectrum (1D, 800 MHz, D2O, predicted) | 2021-09-24 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 13C NMR Spectrum (1D, 900 MHz, D2O, predicted) | 2021-09-24 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 1H NMR Spectrum (1D, 900 MHz, D2O, predicted) | 2021-09-24 | Wishart Lab | View Spectrum |
|
|---|