| Record Information |
|---|
| Version | 5.0 |
|---|
| Status | Detected and Quantified |
|---|
| Creation Date | 2005-11-16 15:48:42 UTC |
|---|
| Update Date | 2021-09-14 14:57:20 UTC |
|---|
| HMDB ID | HMDB0000217 |
|---|
| Secondary Accession Numbers | |
|---|
| Metabolite Identification |
|---|
| Common Name | NADP |
|---|
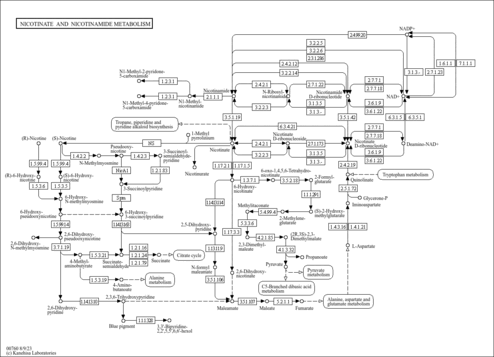
| Description | Nicotinamide adenine dinucleotide phosphate (NADP) is a coenzyme composed of ribosylnicotinamide 5'-phosphate (NMN) coupled with a pyrophosphate linkage to 5'-phosphate adenosine 2',5'-bisphosphate. NADP serves as an electron carrier in a number of reactions, being alternately oxidized (NADP+) and reduced (NADPH). NADP is formed through the addition of a phosphate group to the 2' position of the adenosyl nucleotide through an ester linkage (Dorland, 27th ed). This extra phosphate is added by the enzyme NAD+ kinase and removed via NADP+ phosphatase. NADP is also known as TPN (triphosphopyridine nucleotide) and it is an important cofactor used in anabolic reactions in all forms of cellular life. Examples include the Calvin cycle, cholesterol synthesis, fatty acid elongation, and nucleic acid synthesis (Wikipedia ). |
|---|
| Structure | NC(=O)C1=C[N+](=CC=C1)[C@@H]1O[C@H](COP([O-])(=O)OP(O)(=O)OC[C@H]2O[C@H]([C@H](OP(O)(O)=O)[C@@H]2O)N2C=NC3=C2N=CN=C3N)[C@@H](O)[C@H]1O InChI=1S/C21H28N7O17P3/c22-17-12-19(25-7-24-17)28(8-26-12)21-16(44-46(33,34)35)14(30)11(43-21)6-41-48(38,39)45-47(36,37)40-5-10-13(29)15(31)20(42-10)27-3-1-2-9(4-27)18(23)32/h1-4,7-8,10-11,13-16,20-21,29-31H,5-6H2,(H7-,22,23,24,25,32,33,34,35,36,37,38,39)/t10-,11-,13-,14-,15-,16-,20-,21-/m1/s1 |
|---|
| Synonyms | | Value | Source |
|---|
| Codehydrase II | ChEBI | | Codehydrogenase II | ChEBI | | Coenzyme II | ChEBI | | NAD phosphate | ChEBI | | NADP nicotinamide-adenine-dinucleotide phosphATE | ChEBI | | TPN | ChEBI | | Triphosphopyridine nucleotide | ChEBI | | NAD phosphoric acid | Generator | | NADP nicotinamide-adenine-dinucleotide phosphoric acid | Generator | | NADPH | MeSH | | Nicotinamide-adenine dinucleotide phosphate | MeSH | | Nucleotide, triphosphopyridine | MeSH | | Dinucleotide phosphate, nicotinamide-adenine | MeSH | | Nicotinamide adenine dinucleotide phosphate | MeSH | | Phosphate, nicotinamide-adenine dinucleotide | MeSH | | NADP | ChEBI, MeSH | | Adenine-nicotinamide dinucleotide phosphate | HMDB | | NADP+ | HMDB | | beta-NADP | HMDB | | beta-Nicotinamide adenine dinucleotide phosphate | HMDB | | beta-TPN | HMDB | | β-NADP | HMDB | | β-Nicotinamide adenine dinucleotide phosphate | HMDB | | β-TPN | HMDB |
|
|---|
| Chemical Formula | C21H28N7O17P3 |
|---|
| Average Molecular Weight | 743.405 |
|---|
| Monoisotopic Molecular Weight | 743.075452041 |
|---|
| IUPAC Name | 1-[(2R,3R,4S,5R)-5-{[({[(2R,3R,4R,5R)-5-(6-amino-9H-purin-9-yl)-3-hydroxy-4-(phosphonooxy)oxolan-2-yl]methoxy}(hydroxy)phosphoryl phosphono)oxy]methyl}-3,4-dihydroxyoxolan-2-yl]-3-carbamoyl-1lambda5-pyridin-1-ylium |
|---|
| Traditional Name | 1-[(2R,3R,4S,5R)-5-[({[(2R,3R,4R,5R)-5-(6-aminopurin-9-yl)-3-hydroxy-4-(phosphonooxy)oxolan-2-yl]methoxy(hydroxy)phosphoryl phosphono}oxy)methyl]-3,4-dihydroxyoxolan-2-yl]-3-carbamoyl-1lambda5-pyridin-1-ylium |
|---|
| CAS Registry Number | 53-59-8 |
|---|
| SMILES | NC(=O)C1=C[N+](=CC=C1)[C@@H]1O[C@H](COP([O-])(=O)OP(O)(=O)OC[C@H]2O[C@H]([C@H](OP(O)(O)=O)[C@@H]2O)N2C=NC3=C2N=CN=C3N)[C@@H](O)[C@H]1O |
|---|
| InChI Identifier | InChI=1S/C21H28N7O17P3/c22-17-12-19(25-7-24-17)28(8-26-12)21-16(44-46(33,34)35)14(30)11(43-21)6-41-48(38,39)45-47(36,37)40-5-10-13(29)15(31)20(42-10)27-3-1-2-9(4-27)18(23)32/h1-4,7-8,10-11,13-16,20-21,29-31H,5-6H2,(H7-,22,23,24,25,32,33,34,35,36,37,38,39)/t10-,11-,13-,14-,15-,16-,20-,21-/m1/s1 |
|---|
| InChI Key | XJLXINKUBYWONI-NNYOXOHSSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | Belongs to the class of organic compounds known as (5'->5')-dinucleotides. These are dinucleotides where the two bases are connected via a (5'->5')-phosphodiester linkage. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Nucleosides, nucleotides, and analogues |
|---|
| Class | (5'->5')-dinucleotides |
|---|
| Sub Class | Not Available |
|---|
| Direct Parent | (5'->5')-dinucleotides |
|---|
| Alternative Parents | |
|---|
| Substituents | - (5'->5')-dinucleotide
- Purine nucleotide sugar
- Purine ribonucleoside 2',5'-bisphosphate
- Purine ribonucleoside bisphosphate
- Purine ribonucleoside diphosphate
- Nicotinamide-nucleotide
- Pyridine nucleotide
- Pentose phosphate
- Pentose-5-phosphate
- Glycosyl compound
- N-glycosyl compound
- 6-aminopurine
- Monosaccharide phosphate
- Organic pyrophosphate
- Imidazopyrimidine
- Nicotinamide
- Purine
- Aminopyrimidine
- Monoalkyl phosphate
- Monosaccharide
- N-substituted imidazole
- Organic phosphoric acid derivative
- Pyrimidine
- Alkyl phosphate
- Phosphoric acid ester
- Pyridinium
- Pyridine
- Imidolactam
- Imidazole
- Tetrahydrofuran
- Heteroaromatic compound
- Azole
- Secondary alcohol
- Carboximidic acid derivative
- Oxacycle
- Azacycle
- Organoheterocyclic compound
- Carboximidic acid
- Primary amine
- Alcohol
- Organopnictogen compound
- Organic oxygen compound
- Organic nitrogen compound
- Amine
- Organonitrogen compound
- Organooxygen compound
- Organic oxide
- Organic zwitterion
- Hydrocarbon derivative
- Aromatic heteropolycyclic compound
|
|---|
| Molecular Framework | Aromatic heteropolycyclic compounds |
|---|
| External Descriptors | |
|---|
| Ontology |
|---|
| Not Available | Not Available |
|---|
| Physical Properties |
|---|
| State | Solid |
|---|
| Experimental Molecular Properties | | Property | Value | Reference |
|---|
| Melting Point | Not Available | Not Available | | Boiling Point | Not Available | Not Available | | Water Solubility | Not Available | Not Available | | LogP | Not Available | Not Available |
|
|---|
| Experimental Chromatographic Properties | Not Available |
|---|
| Predicted Molecular Properties | |
|---|
| Predicted Chromatographic Properties | Predicted Collision Cross SectionsPredicted Retention Times Underivatized| Chromatographic Method | Retention Time | Reference |
|---|
| Measured using a Waters Acquity ultraperformance liquid chromatography (UPLC) ethylene-bridged hybrid (BEH) C18 column (100 mm × 2.1 mm; 1.7 μmparticle diameter). Predicted by Afia on May 17, 2022. Predicted by Afia on May 17, 2022. | 2.7 minutes | 32390414 | | Predicted by Siyang on May 30, 2022 | 10.1591 minutes | 33406817 | | Predicted by Siyang using ReTip algorithm on June 8, 2022 | 9.31 minutes | 32390414 | | AjsUoB = Accucore 150 Amide HILIC with 10mM Ammonium Formate, 0.1% Formic Acid | 546.0 seconds | 40023050 | | Fem_Long = Waters ACQUITY UPLC HSS T3 C18 with Water:MeOH and 0.1% Formic Acid | 697.3 seconds | 40023050 | | Fem_Lipids = Ascentis Express C18 with (60:40 water:ACN):(90:10 IPA:ACN) and 10mM NH4COOH + 0.1% Formic Acid | 196.9 seconds | 40023050 | | Life_Old = Waters ACQUITY UPLC BEH C18 with Water:(20:80 acetone:ACN) and 0.1% Formic Acid | 58.5 seconds | 40023050 | | Life_New = RP Waters ACQUITY UPLC HSS T3 C18 with Water:(30:70 MeOH:ACN) and 0.1% Formic Acid | 165.8 seconds | 40023050 | | RIKEN = Waters ACQUITY UPLC BEH C18 with Water:ACN and 0.1% Formic Acid | 63.5 seconds | 40023050 | | Eawag_XBridgeC18 = XBridge C18 3.5u 2.1x50 mm with Water:MeOH and 0.1% Formic Acid | 357.4 seconds | 40023050 | | BfG_NTS_RP1 =Agilent Zorbax Eclipse Plus C18 (2.1 mm x 150 mm, 3.5 um) with Water:ACN and 0.1% Formic Acid | 351.8 seconds | 40023050 | | HILIC_BDD_2 = Merck SeQuant ZIC-HILIC with ACN(0.1% formic acid):water(16 mM ammonium formate) | 1080.8 seconds | 40023050 | | UniToyama_Atlantis = RP Waters Atlantis T3 (2.1 x 150 mm, 5 um) with ACN:Water and 0.1% Formic Acid | 611.8 seconds | 40023050 | | BDD_C18 = Hypersil Gold 1.9µm C18 with Water:ACN and 0.1% Formic Acid | 86.4 seconds | 40023050 | | UFZ_Phenomenex = Kinetex Core-Shell C18 2.6 um, 3.0 x 100 mm, Phenomenex with Water:MeOH and 0.1% Formic Acid | 668.5 seconds | 40023050 | | SNU_RIKEN_POS = Waters ACQUITY UPLC BEH C18 with Water:ACN and 0.1% Formic Acid | 183.6 seconds | 40023050 | | RPMMFDA = Waters ACQUITY UPLC BEH C18 with Water:ACN and 0.1% Formic Acid | 279.6 seconds | 40023050 | | MTBLS87 = Merck SeQuant ZIC-pHILIC column with ACN:Water and :ammonium carbonate | 662.9 seconds | 40023050 | | KI_GIAR_zic_HILIC_pH2_7 = Merck SeQuant ZIC-HILIC with ACN:Water and 0.1% FA | 447.8 seconds | 40023050 | | Meister zic-pHILIC pH9.3 = Merck SeQuant ZIC-pHILIC column with ACN:Water 5mM NH4Ac pH9.3 and 5mM ammonium acetate in water | 598.5 seconds | 40023050 |
Predicted Kovats Retention IndicesUnderivatized |
|---|