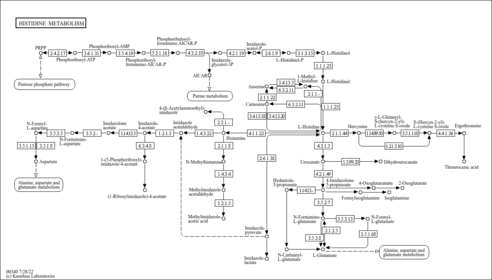
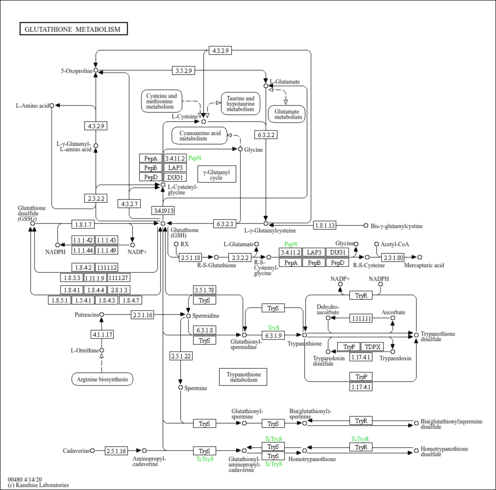
Showing metabocard for NADPH (HMDB0000221)
Only showing the first 10 proteins. There are 203 proteins in total.
Enzymes
- General function:
- Involved in transferase activity
- Specific function:
- Fatty acid synthetase catalyzes the formation of long-chain fatty acids from acetyl-CoA, malonyl-CoA and NADPH. This multifunctional protein has 7 catalytic activities and an acyl carrier protein.
- Gene Name:
- FASN
- Uniprot ID:
- P49327
- Molecular weight:
- 273424.06
Reactions
| (3R)-3-hydroxyacyl-[acyl-carrier-protein] + NADP → 3-oxoacyl-[acyl-carrier-protein] + NADPH | details |
| Acyl-[acyl-carrier-protein] + NADP → trans-2,3-dehydroacyl-[acyl-carrier-protein] + NADPH | details |
| Butyryl-[acp] + NADP → But-2-enoyl-[acyl-carrier protein] + NADPH + Hydrogen Ion | details |
| (3R)-3-Hydroxybutanoyl-[acyl-carrier protein] + NADP → Acetoacetyl-[acp] + NADPH + Hydrogen Ion | details |
| (3R)-3-Hydroxydecanoyl-[acyl-carrier protein] + NADP → 3-Oxodecanoyl-[acp] + NADPH | details |
| (3R)-3-Hydroxyoctanoyl-[acyl-carrier protein] + NADP → 3-Oxooctanoyl-[acp] + NADPH + Hydrogen Ion | details |
| (3R)-3-Hydroxypalmitoyl-[acyl-carrier protein] + NADP → 3-Oxohexadecanoyl-[acp] + NADPH + Hydrogen Ion | details |
| (3R)-3-Hydroxytetradecanoyl-[acyl-carrier protein] + NADP → 3-Oxotetradecanoyl-[acp] + NADPH + Hydrogen Ion | details |
| Dodecanoyl-[acyl-carrier protein] + NADP → trans-Dodec-2-enoyl-[acp] + NADPH + Hydrogen Ion | details |
| (R)-3-Hydroxyhexanoyl-[acp] + NADP → 3-Oxohexanoyl-[acp] + NADPH + Hydrogen Ion | details |
| Hexanoyl-[acp] + NADP → trans-Hex-2-enoyl-[acp] + NADPH + Hydrogen Ion | details |
| Octanoyl-[acp] + NADP → trans-Oct-2-enoyl-[acp] + NADPH + Hydrogen Ion | details |
| Decanoyl-[acp] + NADP → trans-Dec-2-enoyl-[acp] + NADPH + Hydrogen Ion | details |
| Tetradecanoyl-[acp] + NADP → trans-Tetradec-2-enoyl-[acp] + NADPH + Hydrogen Ion | details |
| Hexadecanoyl-[acp] + NADP → trans-Hexadec-2-enoyl-[acp] + NADPH + Hydrogen Ion | details |
- General function:
- Involved in oxidoreductase activity
- Specific function:
- Plays a role in valine and pyrimidine metabolism. Binds fatty acyl-CoA.
- Gene Name:
- ALDH6A1
- Uniprot ID:
- Q02252
- Molecular weight:
- 57839.31
Reactions
| Malonic semialdehyde + Coenzyme A + NADP → Acetyl-CoA + Carbon dioxide + NADPH + Hydrogen Ion | details |
| Malonic semialdehyde + Coenzyme A + NADP → Malonyl-CoA + NADPH + Hydrogen Ion | details |
- General function:
- Involved in oxidoreductase activity
- Specific function:
- Catalyzes the transformation of the potent androgen dihydrotestosterone (DHT) into the less active form, 5-alpha-androstan-3-alpha,17-beta-diol (3-alpha-diol). Also has some 20-alpha-hydroxysteroid dehydrogenase activity. The biotransformation of the pesticide chlordecone (kepone) to its corresponding alcohol leads to increased biliary excretion of the pesticide and concomitant reduction of its neurotoxicity since bile is the major excretory route.
- Gene Name:
- AKR1C4
- Uniprot ID:
- P17516
- Molecular weight:
- 37094.57
Reactions
| Chlordecone alcohol + NADP → Chlordecone + NADPH | details |
| Androsterone + NADP → Androstanedione + NADPH + Hydrogen Ion | details |
| Etiocholanolone + NADP → Etiocholanedione + NADPH + Hydrogen Ion | details |
| 3a,7a-Dihydroxy-5b-cholestane + NADP → 7a-Hydroxy-5b-cholestan-3-one + NADPH + Hydrogen Ion | details |
| 5beta-Cholestane-3alpha,7alpha,12alpha-triol + NADP → 7a,12a-Dihydroxy-5b-cholestan-3-one + NADPH + Hydrogen Ion | details |
| 17a,21-Dihydroxy-5b-pregnane-3,11,20-trione + Hydrogen Ion + NADPH → Tetrahydrocortisone + NADP | details |
| Tetrahydrocortisol + NADP → Dihydrocortisol + NADPH + Hydrogen Ion | details |
| 3a,11b,21-Trihydroxy-20-oxo-5b-pregnan-18-al + NADP → 11b,21-Dihydroxy-3,20-oxo-5b-pregnan-18-al + NADPH + Hydrogen Ion | details |
| Tetrahydrocorticosterone + NADP → 11b,21-Dihydroxy-5b-pregnane-3,20-dione + NADPH + Hydrogen Ion | details |
| 3a,21-Dihydroxy-5b-pregnane-11,20-dione + NADP → 21-Hydroxy-5b-pregnane-3,11,20-trione + NADPH + Hydrogen Ion | details |
| 3a-Hydroxy-5b-pregnane-20-one + NADP → 5a-Pregnane-3,20-dione + NADPH + Hydrogen Ion | details |
| Dihydrotestosterone + NADPH + Hydrogen Ion → Androstan-3alpha,17beta-diol + NADP | details |
- General function:
- Involved in oxidoreductase activity
- Specific function:
- NADPH-dependent reductase with broad substrate specificity. Catalyzes the reduction of a wide variety of carbonyl compounds including quinones, prostaglandins, menadione, plus various xenobiotics. Catalyzes the reduction of the antitumor anthracyclines doxorubicin and daunorubicin to the cardiotoxic compounds doxorubicinol and daunorubicinol. Can convert prostaglandin E2 to prostaglandin F2-alpha. Can bind glutathione, which explains its higher affinity for glutathione-conjugated substrates. Catalyzes the reduction of S-nitrosoglutathione.
- Gene Name:
- CBR1
- Uniprot ID:
- P16152
- Molecular weight:
- 30374.73
Reactions
| R-CHOH-R' + NADP → R-CO-R' + NADPH | details |
| (5Z,13E)-(15S)-9-alpha,11-alpha,15-trihydroxyprosta-5,13-dienoate + NADP → (5Z,13E)-(15S)-11-alpha,15-dihydroxy-9-oxoprosta-5,13-dienoate + NADPH | details |
| (13E)-(15S)-11-alpha,15-dihydroxy-9-oxoprost-13-enoate + NADP → (13E)-11-alpha-hydroxy-9,15-dioxoprost-13-enoate + NADPH | details |
| Prostaglandin F2a + NADP → Prostaglandin E2 + NADPH + Hydrogen Ion | details |
| 4-(Methylnitrosamino)-1-(3-pyridyl)-1-butanone + NADPH + Hydrogen Ion → 4-(Methylnitrosamino)-1-(3-pyridyl)-1-butanol + NADP | details |
- General function:
- Involved in oxidoreductase activity
- Specific function:
- Reduces all-trans-retinal and 9-cis retinal. Can also catalyze the oxidation of all-trans-retinol with NADP as co-factor, but with much lower efficiency. Reduces alkyl phenyl ketones and alpha-dicarbonyl compounds with aromatic rings, such as pyrimidine-4-aldehyde, 3-benzoylpyridine, 4-benzoylpyridine, menadione and 4-hexanoylpyridine. Has no activity towards aliphatic aldehydes and ketones (By similarity).
- Gene Name:
- DHRS4
- Uniprot ID:
- Q9BTZ2
- Molecular weight:
- 29536.885
Reactions
| R-CHOH-R' + NADP → R-CO-R' + NADPH | details |
- General function:
- Involved in oxidoreductase activity
- Specific function:
- Has low NADPH-dependent oxidoreductase activity towards 4-benzoylpyridine and menadione (in vitro).
- Gene Name:
- CBR3
- Uniprot ID:
- O75828
- Molecular weight:
- 30849.97
Reactions
| R-CHOH-R' + NADP → R-CO-R' + NADPH | details |
| Prostaglandin F2a + NADP → Prostaglandin E2 + NADPH + Hydrogen Ion | details |
| 4-(Methylnitrosamino)-1-(3-pyridyl)-1-butanone + NADPH + Hydrogen Ion → 4-(Methylnitrosamino)-1-(3-pyridyl)-1-butanol + NADP | details |
- General function:
- Involved in oxidoreductase activity
- Specific function:
- Catalyzes the NADPH-dependent reduction of a wide variety of carbonyl-containing compounds to their corresponding alcohols with a broad range of catalytic efficiencies.
- Gene Name:
- AKR1B1
- Uniprot ID:
- P15121
- Molecular weight:
- 35853.125
Reactions
| Glycerol + NADP → Glyceraldehyde + NADPH + Hydrogen Ion | details |
| beta-D-Galactose + NADPH + Hydrogen Ion → Galactitol + NADP | details |
| D-Xylitol + NADP → D-Xylose + NADPH + Hydrogen Ion | details |
| L-Arabitol + NADP → L-Arabinose + NADPH + Hydrogen Ion | details |
| Sorbitol + NADP → alpha-D-Glucose + NADPH + Hydrogen Ion | details |
| Propylene glycol + NADP → Lactaldehyde + NADPH + Hydrogen Ion | details |
- General function:
- Involved in oxidoreductase activity
- Specific function:
- Isoform 1 may possess glutaredoxin activity as well as thioredoxin reductase activity and induces actin and tubulin polymerization, leading to formation of cell membrane protrusions. Isoform 4 enhances the transcriptional activity of estrogen receptors alpha and beta while isoform 5 enhances the transcriptional activity of the beta receptor only. Isoform 5 also mediates cell death induced by a combination of interferon-beta and retinoic acid.
- Gene Name:
- TXNRD1
- Uniprot ID:
- Q16881
- Molecular weight:
- 70905.58
Reactions
| Thioredoxin + NADP → thioredoxin disulfide + NADPH | details |
| Thioredoxin + NADP → Thioredoxin disulfide + NADPH + Hydrogen Ion | details |
| Hydrogen selenide + NADP + Water → Selenite + NADPH + Hydrogen Ion | details |
| NADPH + Hydrogen Ion + Methylselenic acid → NADP + Water + Methaneselenol | details |
- General function:
- Involved in iron ion binding
- Specific function:
- Catalyzes a dehydrogenation to introduce C5-6 double bond into lathosterol.
- Gene Name:
- SC5DL
- Uniprot ID:
- O75845
- Molecular weight:
- 35300.55
Reactions
| Lathosterol + NADPH + Hydrogen Ion + Oxygen → 7-Dehydrocholesterol + NADP + Water | details |
| Episterol + NADP → 5,7,24(28)-Ergostatrienol + NADPH + Hydrogen Ion | details |
- General function:
- Involved in flavin-containing monooxygenase activity
- Specific function:
- In contrast with other forms of FMO it does not seem to be a drug-metabolizing enzyme.
- Gene Name:
- FMO5
- Uniprot ID:
- P49326
- Molecular weight:
- 32480.04
Reactions
| N,N-Dimethylaniline + NADPH + Oxygen → Dimethylaniline-N-oxide + NADP + Water | details |
| Trimethylamine + NADPH + Hydrogen Ion + Oxygen → Trimethylamine N-oxide + NADP + Water | details |
| Tamoxifen + Oxygen + NADPH + Hydrogen Ion → Tamoxifen N-oxide + NADP + Water | details |
Only showing the first 10 proteins. There are 203 proteins in total.