| Record Information |
|---|
| Version | 5.0 |
|---|
| Status | Detected and Quantified |
|---|
| Creation Date | 2005-11-16 15:48:42 UTC |
|---|
| Update Date | 2023-02-21 17:15:29 UTC |
|---|
| HMDB ID | HMDB0001149 |
|---|
| Secondary Accession Numbers | |
|---|
| Metabolite Identification |
|---|
| Common Name | 5-Aminolevulinic acid |
|---|
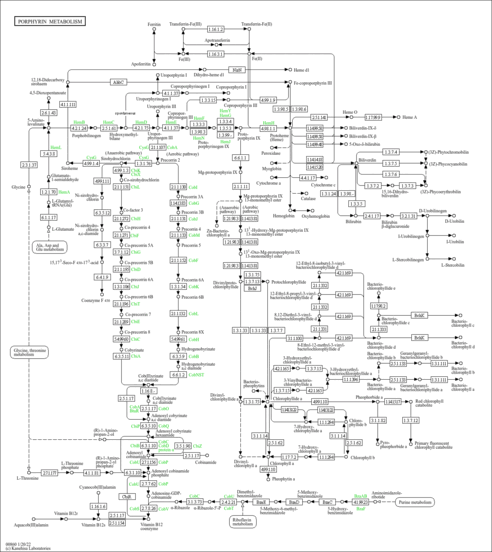
| Description | 5-Aminolevulinic acid, also known as 5-aminolevulinate or 5-amino-4-oxopentanoate, belongs to the class of organic compounds known as delta amino acids and derivatives. Delta amino acids and derivatives are compounds containing a carboxylic acid group and an amino group at the C5 carbon atom. 5-Aminolevulinic acid is a very hydrophobic molecule, practically insoluble in water, and relatively neutral. 5-Aminolevulinic acid exists in all living species, ranging from bacteria to humans. 5-aminolevulinic acid can be biosynthesized from glycine and succinyl-CoA by the enzyme 5-aminolevulinate synthase. The simplest delta-amino acid in which the hydrogens at the gamma position are replaced by an oxo group. In humans, 5-aminolevulinic acid is involved in the metabolic disorder called the dimethylglycine dehydrogenase deficiency pathway. Outside of the human body, 5-Aminolevulinic acid has been detected, but not quantified in several different foods, such as american butterfish, vaccinium (blueberry, cranberry, huckleberry), amaranths, purple mangosteens, and garden cress. Used (in the form of the hydrochloride salt) in combination with blue light illumination for the treatment of minimally to moderately thick actinic keratosis of the face or scalp. It is metabolised to protoporphyrin IX, a photoactive compound which accumulates in the skin. |
|---|
| Structure | InChI=1S/C5H9NO3/c6-3-4(7)1-2-5(8)9/h1-3,6H2,(H,8,9) |
|---|
| Synonyms | | Value | Source |
|---|
| 5-ALA | ChEBI | | 5-Amino-4-oxopentanoate | ChEBI | | 5-Amino-4-oxovaleric acid | ChEBI | | 5-Aminolevulinate | ChEBI | | Aminolevulinic acid | ChEBI | | DALA | ChEBI | | delta-ALA | ChEBI | | delta-Aminolevulinic acid | ChEBI | | 5-Amino-4-oxopentanoic acid | Generator | | 5-Amino-4-oxovalerate | Generator | | Aminolevulinate | Generator | | Δ-ala | Generator | | delta-Aminolevulinate | Generator | | Δ-aminolevulinate | Generator | | Δ-aminolevulinic acid | Generator | | 5-Amino-4-oxo-pentanoate | HMDB | | 5-Amino-4-oxo-pentanoic acid | HMDB | | 5-Amino-levulinate | HMDB | | 5-Amino-levulinic acid | HMDB | | 5-Aminolaevulinate | HMDB | | 5-Aminolaevulinic acid | HMDB | | Aladerm | HMDB | | Amino-levulinic acid | HMDB | | Kerastick | HMDB | | 5 Aminolevulinate | HMDB | | Acid, Delta-aminolevulinic | HMDB | | Levulan | HMDB | | 5 Aminolaevulinate | HMDB | | Acid hydrochloride, aminolevulinic | HMDB | | Bertek brand OF aminolevulinic acid hydrochloride | HMDB | | Hydrochloride, aminolevulinic acid | HMDB | | Acid, aminolevulinic | HMDB | | Aminolevulinic acid hydrochloride | HMDB | | Delta Aminolevulinic acid | HMDB |
|
|---|
| Chemical Formula | C5H9NO3 |
|---|
| Average Molecular Weight | 131.1299 |
|---|
| Monoisotopic Molecular Weight | 131.058243159 |
|---|
| IUPAC Name | 5-amino-4-oxopentanoic acid |
|---|
| Traditional Name | aminolevulinic acid |
|---|
| CAS Registry Number | 106-60-5 |
|---|
| SMILES | NCC(=O)CCC(O)=O |
|---|
| InChI Identifier | InChI=1S/C5H9NO3/c6-3-4(7)1-2-5(8)9/h1-3,6H2,(H,8,9) |
|---|
| InChI Key | ZGXJTSGNIOSYLO-UHFFFAOYSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | Belongs to the class of organic compounds known as delta amino acids and derivatives. Delta amino acids and derivatives are compounds containing a carboxylic acid group and an amino group at the C5 carbon atom. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Organic acids and derivatives |
|---|
| Class | Carboxylic acids and derivatives |
|---|
| Sub Class | Amino acids, peptides, and analogues |
|---|
| Direct Parent | Delta amino acids and derivatives |
|---|
| Alternative Parents | |
|---|
| Substituents | - Delta amino acid or derivatives
- Gamma-keto acid
- Short-chain keto acid
- Keto acid
- Alpha-aminoketone
- Amino acid
- Ketone
- Carboxylic acid
- Monocarboxylic acid or derivatives
- Organonitrogen compound
- Organic nitrogen compound
- Primary aliphatic amine
- Hydrocarbon derivative
- Organic oxide
- Organopnictogen compound
- Organic oxygen compound
- Carbonyl group
- Amine
- Organooxygen compound
- Primary amine
- Aliphatic acyclic compound
|
|---|
| Molecular Framework | Aliphatic acyclic compounds |
|---|
| External Descriptors | |
|---|
| Ontology |
|---|
| Physiological effect | Not Available |
|---|
| Disposition | |
|---|
| Process | |
|---|
| Role | |
|---|
| Physical Properties |
|---|
| State | Solid |
|---|
| Experimental Molecular Properties | | Property | Value | Reference |
|---|
| Melting Point | 156 - 158 °C | Not Available | | Boiling Point | Not Available | Not Available | | Water Solubility | 1000 mg/mL at 25 °C | Not Available | | LogP | Not Available | Not Available |
|
|---|
| Experimental Chromatographic Properties | Experimental Collision Cross Sections |
|---|
| Predicted Molecular Properties | |
|---|
| Predicted Chromatographic Properties | Predicted Collision Cross SectionsPredicted Kovats Retention IndicesUnderivatizedDerivatized| Derivative Name / Structure | SMILES | Kovats RI Value | Column Type | Reference |
|---|
| 5-Aminolevulinic acid,1TMS,isomer #1 | C[Si](C)(C)OC(=O)CCC(=O)CN | 1389.2 | Semi standard non polar | 33892256 | | 5-Aminolevulinic acid,1TMS,isomer #2 | C[Si](C)(C)OC(=CCC(=O)O)CN | 1473.0 | Semi standard non polar | 33892256 | | 5-Aminolevulinic acid,1TMS,isomer #3 | C[Si](C)(C)OC(=CN)CCC(=O)O | 1507.8 | Semi standard non polar | 33892256 | | 5-Aminolevulinic acid,1TMS,isomer #4 | C[Si](C)(C)NCC(=O)CCC(=O)O | 1433.3 | Semi standard non polar | 33892256 | | 5-Aminolevulinic acid,2TMS,isomer #1 | C[Si](C)(C)OC(=O)CCC(=CN)O[Si](C)(C)C | 1522.2 | Semi standard non polar | 33892256 | | 5-Aminolevulinic acid,2TMS,isomer #1 | C[Si](C)(C)OC(=O)CCC(=CN)O[Si](C)(C)C | 1519.5 | Standard non polar | 33892256 | | 5-Aminolevulinic acid,2TMS,isomer #1 | C[Si](C)(C)OC(=O)CCC(=CN)O[Si](C)(C)C | 2207.2 | Standard polar | 33892256 | | 5-Aminolevulinic acid,2TMS,isomer #2 | C[Si](C)(C)OC(=O)CC=C(CN)O[Si](C)(C)C | 1517.2 | Semi standard non polar | 33892256 | | 5-Aminolevulinic acid,2TMS,isomer #2 | C[Si](C)(C)OC(=O)CC=C(CN)O[Si](C)(C)C | 1583.6 | Standard non polar | 33892256 | | 5-Aminolevulinic acid,2TMS,isomer #2 | C[Si](C)(C)OC(=O)CC=C(CN)O[Si](C)(C)C | 2172.5 | Standard polar | 33892256 | | 5-Aminolevulinic acid,2TMS,isomer #3 | C[Si](C)(C)NCC(=O)CCC(=O)O[Si](C)(C)C | 1530.6 | Semi standard non polar | 33892256 | | 5-Aminolevulinic acid,2TMS,isomer #3 | C[Si](C)(C)NCC(=O)CCC(=O)O[Si](C)(C)C | 1555.6 | Standard non polar | 33892256 | | 5-Aminolevulinic acid,2TMS,isomer #3 | C[Si](C)(C)NCC(=O)CCC(=O)O[Si](C)(C)C | 1826.2 | Standard polar | 33892256 | | 5-Aminolevulinic acid,2TMS,isomer #4 | C[Si](C)(C)NCC(=CCC(=O)O)O[Si](C)(C)C | 1605.6 | Semi standard non polar | 33892256 | | 5-Aminolevulinic acid,2TMS,isomer #4 | C[Si](C)(C)NCC(=CCC(=O)O)O[Si](C)(C)C | 1678.5 | Standard non polar | 33892256 | | 5-Aminolevulinic acid,2TMS,isomer #4 | C[Si](C)(C)NCC(=CCC(=O)O)O[Si](C)(C)C | 2022.6 | Standard polar | 33892256 | | 5-Aminolevulinic acid,2TMS,isomer #5 | C[Si](C)(C)NC=C(CCC(=O)O)O[Si](C)(C)C | 1668.6 | Semi standard non polar | 33892256 | | 5-Aminolevulinic acid,2TMS,isomer #5 | C[Si](C)(C)NC=C(CCC(=O)O)O[Si](C)(C)C | 1661.6 | Standard non polar | 33892256 | | 5-Aminolevulinic acid,2TMS,isomer #5 | C[Si](C)(C)NC=C(CCC(=O)O)O[Si](C)(C)C | 1980.5 | Standard polar | 33892256 | | 5-Aminolevulinic acid,2TMS,isomer #6 | C[Si](C)(C)N(CC(=O)CCC(=O)O)[Si](C)(C)C | 1687.0 | Semi standard non polar | 33892256 | | 5-Aminolevulinic acid,2TMS,isomer #6 | C[Si](C)(C)N(CC(=O)CCC(=O)O)[Si](C)(C)C | 1625.0 | Standard non polar | 33892256 | | 5-Aminolevulinic acid,2TMS,isomer #6 | C[Si](C)(C)N(CC(=O)CCC(=O)O)[Si](C)(C)C | 2004.9 | Standard polar | 33892256 | | 5-Aminolevulinic acid,3TMS,isomer #1 | C[Si](C)(C)NC=C(CCC(=O)O[Si](C)(C)C)O[Si](C)(C)C | 1700.3 | Semi standard non polar | 33892256 | | 5-Aminolevulinic acid,3TMS,isomer #1 | C[Si](C)(C)NC=C(CCC(=O)O[Si](C)(C)C)O[Si](C)(C)C | 1671.9 | Standard non polar | 33892256 | | 5-Aminolevulinic acid,3TMS,isomer #1 | C[Si](C)(C)NC=C(CCC(=O)O[Si](C)(C)C)O[Si](C)(C)C | 1777.3 | Standard polar | 33892256 | | 5-Aminolevulinic acid,3TMS,isomer #2 | C[Si](C)(C)NCC(=CCC(=O)O[Si](C)(C)C)O[Si](C)(C)C | 1637.2 | Semi standard non polar | 33892256 | | 5-Aminolevulinic acid,3TMS,isomer #2 | C[Si](C)(C)NCC(=CCC(=O)O[Si](C)(C)C)O[Si](C)(C)C | 1707.7 | Standard non polar | 33892256 | | 5-Aminolevulinic acid,3TMS,isomer #2 | C[Si](C)(C)NCC(=CCC(=O)O[Si](C)(C)C)O[Si](C)(C)C | 1692.4 | Standard polar | 33892256 | | 5-Aminolevulinic acid,3TMS,isomer #3 | C[Si](C)(C)OC(=O)CCC(=O)CN([Si](C)(C)C)[Si](C)(C)C | 1715.9 | Semi standard non polar | 33892256 | | 5-Aminolevulinic acid,3TMS,isomer #3 | C[Si](C)(C)OC(=O)CCC(=O)CN([Si](C)(C)C)[Si](C)(C)C | 1706.8 | Standard non polar | 33892256 | | 5-Aminolevulinic acid,3TMS,isomer #3 | C[Si](C)(C)OC(=O)CCC(=O)CN([Si](C)(C)C)[Si](C)(C)C | 1721.3 | Standard polar | 33892256 | | 5-Aminolevulinic acid,3TMS,isomer #4 | C[Si](C)(C)OC(=CCC(=O)O)CN([Si](C)(C)C)[Si](C)(C)C | 1827.6 | Semi standard non polar | 33892256 | | 5-Aminolevulinic acid,3TMS,isomer #4 | C[Si](C)(C)OC(=CCC(=O)O)CN([Si](C)(C)C)[Si](C)(C)C | 1788.6 | Standard non polar | 33892256 | | 5-Aminolevulinic acid,3TMS,isomer #4 | C[Si](C)(C)OC(=CCC(=O)O)CN([Si](C)(C)C)[Si](C)(C)C | 1912.1 | Standard polar | 33892256 | | 5-Aminolevulinic acid,3TMS,isomer #5 | C[Si](C)(C)OC(=CN([Si](C)(C)C)[Si](C)(C)C)CCC(=O)O | 1867.8 | Semi standard non polar | 33892256 | | 5-Aminolevulinic acid,3TMS,isomer #5 | C[Si](C)(C)OC(=CN([Si](C)(C)C)[Si](C)(C)C)CCC(=O)O | 1721.9 | Standard non polar | 33892256 | | 5-Aminolevulinic acid,3TMS,isomer #5 | C[Si](C)(C)OC(=CN([Si](C)(C)C)[Si](C)(C)C)CCC(=O)O | 1892.3 | Standard polar | 33892256 | | 5-Aminolevulinic acid,4TMS,isomer #1 | C[Si](C)(C)OC(=O)CCC(=CN([Si](C)(C)C)[Si](C)(C)C)O[Si](C)(C)C | 1838.0 | Semi standard non polar | 33892256 | | 5-Aminolevulinic acid,4TMS,isomer #1 | C[Si](C)(C)OC(=O)CCC(=CN([Si](C)(C)C)[Si](C)(C)C)O[Si](C)(C)C | 1715.2 | Standard non polar | 33892256 | | 5-Aminolevulinic acid,4TMS,isomer #1 | C[Si](C)(C)OC(=O)CCC(=CN([Si](C)(C)C)[Si](C)(C)C)O[Si](C)(C)C | 1743.0 | Standard polar | 33892256 | | 5-Aminolevulinic acid,4TMS,isomer #2 | C[Si](C)(C)OC(=O)CC=C(CN([Si](C)(C)C)[Si](C)(C)C)O[Si](C)(C)C | 1826.7 | Semi standard non polar | 33892256 | | 5-Aminolevulinic acid,4TMS,isomer #2 | C[Si](C)(C)OC(=O)CC=C(CN([Si](C)(C)C)[Si](C)(C)C)O[Si](C)(C)C | 1811.9 | Standard non polar | 33892256 | | 5-Aminolevulinic acid,4TMS,isomer #2 | C[Si](C)(C)OC(=O)CC=C(CN([Si](C)(C)C)[Si](C)(C)C)O[Si](C)(C)C | 1645.3 | Standard polar | 33892256 | | 5-Aminolevulinic acid,1TBDMS,isomer #1 | CC(C)(C)[Si](C)(C)OC(=O)CCC(=O)CN | 1617.3 | Semi standard non polar | 33892256 | | 5-Aminolevulinic acid,1TBDMS,isomer #2 | CC(C)(C)[Si](C)(C)OC(=CCC(=O)O)CN | 1722.4 | Semi standard non polar | 33892256 | | 5-Aminolevulinic acid,1TBDMS,isomer #3 | CC(C)(C)[Si](C)(C)OC(=CN)CCC(=O)O | 1753.1 | Semi standard non polar | 33892256 | | 5-Aminolevulinic acid,1TBDMS,isomer #4 | CC(C)(C)[Si](C)(C)NCC(=O)CCC(=O)O | 1697.4 | Semi standard non polar | 33892256 | | 5-Aminolevulinic acid,2TBDMS,isomer #1 | CC(C)(C)[Si](C)(C)OC(=O)CCC(=CN)O[Si](C)(C)C(C)(C)C | 1956.8 | Semi standard non polar | 33892256 | | 5-Aminolevulinic acid,2TBDMS,isomer #1 | CC(C)(C)[Si](C)(C)OC(=O)CCC(=CN)O[Si](C)(C)C(C)(C)C | 1960.7 | Standard non polar | 33892256 | | 5-Aminolevulinic acid,2TBDMS,isomer #1 | CC(C)(C)[Si](C)(C)OC(=O)CCC(=CN)O[Si](C)(C)C(C)(C)C | 2289.5 | Standard polar | 33892256 | | 5-Aminolevulinic acid,2TBDMS,isomer #2 | CC(C)(C)[Si](C)(C)OC(=O)CC=C(CN)O[Si](C)(C)C(C)(C)C | 1970.3 | Semi standard non polar | 33892256 | | 5-Aminolevulinic acid,2TBDMS,isomer #2 | CC(C)(C)[Si](C)(C)OC(=O)CC=C(CN)O[Si](C)(C)C(C)(C)C | 1964.2 | Standard non polar | 33892256 | | 5-Aminolevulinic acid,2TBDMS,isomer #2 | CC(C)(C)[Si](C)(C)OC(=O)CC=C(CN)O[Si](C)(C)C(C)(C)C | 2242.8 | Standard polar | 33892256 | | 5-Aminolevulinic acid,2TBDMS,isomer #3 | CC(C)(C)[Si](C)(C)NCC(=O)CCC(=O)O[Si](C)(C)C(C)(C)C | 1967.9 | Semi standard non polar | 33892256 | | 5-Aminolevulinic acid,2TBDMS,isomer #3 | CC(C)(C)[Si](C)(C)NCC(=O)CCC(=O)O[Si](C)(C)C(C)(C)C | 1973.3 | Standard non polar | 33892256 | | 5-Aminolevulinic acid,2TBDMS,isomer #3 | CC(C)(C)[Si](C)(C)NCC(=O)CCC(=O)O[Si](C)(C)C(C)(C)C | 2007.1 | Standard polar | 33892256 | | 5-Aminolevulinic acid,2TBDMS,isomer #4 | CC(C)(C)[Si](C)(C)NCC(=CCC(=O)O)O[Si](C)(C)C(C)(C)C | 2090.5 | Semi standard non polar | 33892256 | | 5-Aminolevulinic acid,2TBDMS,isomer #4 | CC(C)(C)[Si](C)(C)NCC(=CCC(=O)O)O[Si](C)(C)C(C)(C)C | 2028.7 | Standard non polar | 33892256 | | 5-Aminolevulinic acid,2TBDMS,isomer #4 | CC(C)(C)[Si](C)(C)NCC(=CCC(=O)O)O[Si](C)(C)C(C)(C)C | 2145.0 | Standard polar | 33892256 | | 5-Aminolevulinic acid,2TBDMS,isomer #5 | CC(C)(C)[Si](C)(C)NC=C(CCC(=O)O)O[Si](C)(C)C(C)(C)C | 2165.0 | Semi standard non polar | 33892256 | | 5-Aminolevulinic acid,2TBDMS,isomer #5 | CC(C)(C)[Si](C)(C)NC=C(CCC(=O)O)O[Si](C)(C)C(C)(C)C | 2008.8 | Standard non polar | 33892256 | | 5-Aminolevulinic acid,2TBDMS,isomer #5 | CC(C)(C)[Si](C)(C)NC=C(CCC(=O)O)O[Si](C)(C)C(C)(C)C | 2131.3 | Standard polar | 33892256 | | 5-Aminolevulinic acid,2TBDMS,isomer #6 | CC(C)(C)[Si](C)(C)N(CC(=O)CCC(=O)O)[Si](C)(C)C(C)(C)C | 2114.5 | Semi standard non polar | 33892256 | | 5-Aminolevulinic acid,2TBDMS,isomer #6 | CC(C)(C)[Si](C)(C)N(CC(=O)CCC(=O)O)[Si](C)(C)C(C)(C)C | 2019.1 | Standard non polar | 33892256 | | 5-Aminolevulinic acid,2TBDMS,isomer #6 | CC(C)(C)[Si](C)(C)N(CC(=O)CCC(=O)O)[Si](C)(C)C(C)(C)C | 2113.8 | Standard polar | 33892256 | | 5-Aminolevulinic acid,3TBDMS,isomer #1 | CC(C)(C)[Si](C)(C)NC=C(CCC(=O)O[Si](C)(C)C(C)(C)C)O[Si](C)(C)C(C)(C)C | 2312.6 | Semi standard non polar | 33892256 | | 5-Aminolevulinic acid,3TBDMS,isomer #1 | CC(C)(C)[Si](C)(C)NC=C(CCC(=O)O[Si](C)(C)C(C)(C)C)O[Si](C)(C)C(C)(C)C | 2236.0 | Standard non polar | 33892256 | | 5-Aminolevulinic acid,3TBDMS,isomer #1 | CC(C)(C)[Si](C)(C)NC=C(CCC(=O)O[Si](C)(C)C(C)(C)C)O[Si](C)(C)C(C)(C)C | 2127.8 | Standard polar | 33892256 | | 5-Aminolevulinic acid,3TBDMS,isomer #2 | CC(C)(C)[Si](C)(C)NCC(=CCC(=O)O[Si](C)(C)C(C)(C)C)O[Si](C)(C)C(C)(C)C | 2273.0 | Semi standard non polar | 33892256 | | 5-Aminolevulinic acid,3TBDMS,isomer #2 | CC(C)(C)[Si](C)(C)NCC(=CCC(=O)O[Si](C)(C)C(C)(C)C)O[Si](C)(C)C(C)(C)C | 2259.0 | Standard non polar | 33892256 | | 5-Aminolevulinic acid,3TBDMS,isomer #2 | CC(C)(C)[Si](C)(C)NCC(=CCC(=O)O[Si](C)(C)C(C)(C)C)O[Si](C)(C)C(C)(C)C | 2071.0 | Standard polar | 33892256 | | 5-Aminolevulinic acid,3TBDMS,isomer #3 | CC(C)(C)[Si](C)(C)OC(=O)CCC(=O)CN([Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C | 2349.8 | Semi standard non polar | 33892256 | | 5-Aminolevulinic acid,3TBDMS,isomer #3 | CC(C)(C)[Si](C)(C)OC(=O)CCC(=O)CN([Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C | 2298.0 | Standard non polar | 33892256 | | 5-Aminolevulinic acid,3TBDMS,isomer #3 | CC(C)(C)[Si](C)(C)OC(=O)CCC(=O)CN([Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C | 2077.9 | Standard polar | 33892256 | | 5-Aminolevulinic acid,3TBDMS,isomer #4 | CC(C)(C)[Si](C)(C)OC(=CCC(=O)O)CN([Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C | 2441.1 | Semi standard non polar | 33892256 | | 5-Aminolevulinic acid,3TBDMS,isomer #4 | CC(C)(C)[Si](C)(C)OC(=CCC(=O)O)CN([Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C | 2341.8 | Standard non polar | 33892256 | | 5-Aminolevulinic acid,3TBDMS,isomer #4 | CC(C)(C)[Si](C)(C)OC(=CCC(=O)O)CN([Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C | 2191.4 | Standard polar | 33892256 | | 5-Aminolevulinic acid,3TBDMS,isomer #5 | CC(C)(C)[Si](C)(C)OC(=CN([Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C)CCC(=O)O | 2481.4 | Semi standard non polar | 33892256 | | 5-Aminolevulinic acid,3TBDMS,isomer #5 | CC(C)(C)[Si](C)(C)OC(=CN([Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C)CCC(=O)O | 2247.5 | Standard non polar | 33892256 | | 5-Aminolevulinic acid,3TBDMS,isomer #5 | CC(C)(C)[Si](C)(C)OC(=CN([Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C)CCC(=O)O | 2181.2 | Standard polar | 33892256 | | 5-Aminolevulinic acid,4TBDMS,isomer #1 | CC(C)(C)[Si](C)(C)OC(=O)CCC(=CN([Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C)O[Si](C)(C)C(C)(C)C | 2657.9 | Semi standard non polar | 33892256 | | 5-Aminolevulinic acid,4TBDMS,isomer #1 | CC(C)(C)[Si](C)(C)OC(=O)CCC(=CN([Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C)O[Si](C)(C)C(C)(C)C | 2445.1 | Standard non polar | 33892256 | | 5-Aminolevulinic acid,4TBDMS,isomer #1 | CC(C)(C)[Si](C)(C)OC(=O)CCC(=CN([Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C)O[Si](C)(C)C(C)(C)C | 2174.8 | Standard polar | 33892256 | | 5-Aminolevulinic acid,4TBDMS,isomer #2 | CC(C)(C)[Si](C)(C)OC(=O)CC=C(CN([Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C)O[Si](C)(C)C(C)(C)C | 2655.5 | Semi standard non polar | 33892256 | | 5-Aminolevulinic acid,4TBDMS,isomer #2 | CC(C)(C)[Si](C)(C)OC(=O)CC=C(CN([Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C)O[Si](C)(C)C(C)(C)C | 2527.3 | Standard non polar | 33892256 | | 5-Aminolevulinic acid,4TBDMS,isomer #2 | CC(C)(C)[Si](C)(C)OC(=O)CC=C(CN([Si](C)(C)C(C)(C)C)[Si](C)(C)C(C)(C)C)O[Si](C)(C)C(C)(C)C | 2117.7 | Standard polar | 33892256 |
|
|---|
| GC-MS Spectra| Spectrum Type | Description | Splash Key | Deposition Date | Source | View |
|---|
| Experimental GC-MS | GC-MS Spectrum - 5-Aminolevulinic acid GC-EI-TOF (Pegasus III TOF-MS system, Leco; GC 6890, Agilent Technologies) (3 TMS) | splash10-0fki-2910000000-12bd38ce6e25c61b8e60 | 2014-06-16 | HMDB team, MONA, MassBank | View Spectrum | | Experimental GC-MS | GC-MS Spectrum - 5-Aminolevulinic acid GC-EI-TOF (Pegasus III TOF-MS system, Leco; GC 6890, Agilent Technologies) (3 TMS) | splash10-00dr-4900000000-c11a861a1638dd2c20d8 | 2014-06-16 | HMDB team, MONA, MassBank | View Spectrum | | Experimental GC-MS | GC-MS Spectrum - 5-Aminolevulinic acid GC-EI-TOF (Pegasus III TOF-MS system, Leco; GC 6890, Agilent Technologies) (Non-derivatized) | splash10-00dr-2911000000-d5b5567862328f5a46cd | 2014-06-16 | HMDB team, MONA, MassBank | View Spectrum | | Experimental GC-MS | GC-MS Spectrum - 5-Aminolevulinic acid GC-EI-TOF (Pegasus III TOF-MS system, Leco; GC 6890, Agilent Technologies) (Non-derivatized) | splash10-00dr-3900000000-538e027ec9932b3f56a5 | 2014-06-16 | HMDB team, MONA, MassBank | View Spectrum | | Experimental GC-MS | GC-MS Spectrum - 5-Aminolevulinic acid GC-EI-TOF (Pegasus III TOF-MS system, Leco; GC 6890, Agilent Technologies) (3 TMS; 1 MEOX) | splash10-00di-9500000000-c0d571fa1aa74cf69ea6 | 2014-06-16 | HMDB team, MONA, MassBank | View Spectrum | | Experimental GC-MS | GC-MS Spectrum - 5-Aminolevulinic acid GC-EI-TOF (Pegasus III TOF-MS system, Leco; GC 6890, Agilent Technologies) (3 TMS; 1 MEOX) | splash10-00di-9600000000-d0a9f31de64870117dfe | 2014-06-16 | HMDB team, MONA, MassBank | View Spectrum | | Experimental GC-MS | GC-MS Spectrum - 5-Aminolevulinic acid GC-MS (1 MEOX; 3 TMS) | splash10-00di-1911000000-117a44ade2fd70812e5c | 2014-06-16 | HMDB team, MONA, MassBank | View Spectrum | | Experimental GC-MS | GC-MS Spectrum - 5-Aminolevulinic acid GC-MS (1 MEOX; 3 TMS) | splash10-00dr-2900000000-635a7d4012b9ef5150f9 | 2014-06-16 | HMDB team, MONA, MassBank | View Spectrum | | Experimental GC-MS | GC-MS Spectrum - 5-Aminolevulinic acid GC-EI-TOF (Non-derivatized) | splash10-0fki-2910000000-12bd38ce6e25c61b8e60 | 2017-09-12 | HMDB team, MONA, MassBank | View Spectrum | | Experimental GC-MS | GC-MS Spectrum - 5-Aminolevulinic acid GC-EI-TOF (Non-derivatized) | splash10-00dr-4900000000-c11a861a1638dd2c20d8 | 2017-09-12 | HMDB team, MONA, MassBank | View Spectrum | | Experimental GC-MS | GC-MS Spectrum - 5-Aminolevulinic acid GC-EI-TOF (Non-derivatized) | splash10-00dr-2911000000-d5b5567862328f5a46cd | 2017-09-12 | HMDB team, MONA, MassBank | View Spectrum | | Experimental GC-MS | GC-MS Spectrum - 5-Aminolevulinic acid GC-EI-TOF (Non-derivatized) | splash10-00dr-3900000000-538e027ec9932b3f56a5 | 2017-09-12 | HMDB team, MONA, MassBank | View Spectrum | | Experimental GC-MS | GC-MS Spectrum - 5-Aminolevulinic acid GC-EI-TOF (Non-derivatized) | splash10-00di-9500000000-c0d571fa1aa74cf69ea6 | 2017-09-12 | HMDB team, MONA, MassBank | View Spectrum | | Experimental GC-MS | GC-MS Spectrum - 5-Aminolevulinic acid GC-EI-TOF (Non-derivatized) | splash10-00di-9600000000-d0a9f31de64870117dfe | 2017-09-12 | HMDB team, MONA, MassBank | View Spectrum | | Experimental GC-MS | GC-MS Spectrum - 5-Aminolevulinic acid GC-MS (Non-derivatized) | splash10-00di-1911000000-117a44ade2fd70812e5c | 2017-09-12 | HMDB team, MONA, MassBank | View Spectrum | | Experimental GC-MS | GC-MS Spectrum - 5-Aminolevulinic acid GC-MS (Non-derivatized) | splash10-00dr-2900000000-635a7d4012b9ef5150f9 | 2017-09-12 | HMDB team, MONA, MassBank | View Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - 5-Aminolevulinic acid GC-MS (Non-derivatized) - 70eV, Positive | splash10-001i-9000000000-b1941f10190ebb6e1343 | 2016-09-22 | Wishart Lab | View Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - 5-Aminolevulinic acid GC-MS (1 TMS) - 70eV, Positive | splash10-05ai-9200000000-6a559cb30668be35a1ed | 2017-10-06 | Wishart Lab | View Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - 5-Aminolevulinic acid GC-MS (Non-derivatized) - 70eV, Positive | Not Available | 2021-10-12 | Wishart Lab | View Spectrum |
MS/MS Spectra| Spectrum Type | Description | Splash Key | Deposition Date | Source | View |
|---|
| Experimental LC-MS/MS | LC-MS/MS Spectrum - 5-Aminolevulinic acid Quattro_QQQ 10V, Positive-QTOF (Annotated) | splash10-0udi-5900000000-cf9a0266243b1a1d0f73 | 2012-07-24 | HMDB team, MONA | View Spectrum | | Experimental LC-MS/MS | LC-MS/MS Spectrum - 5-Aminolevulinic acid Quattro_QQQ 25V, Positive-QTOF (Annotated) | splash10-000i-9000000000-995eb11961b16f254be2 | 2012-07-24 | HMDB team, MONA | View Spectrum | | Experimental LC-MS/MS | LC-MS/MS Spectrum - 5-Aminolevulinic acid Quattro_QQQ 40V, Positive-QTOF (Annotated) | splash10-0fb9-9000000000-64b1ef51cceb8d346f52 | 2012-07-24 | HMDB team, MONA | View Spectrum | | Experimental LC-MS/MS | LC-MS/MS Spectrum - 5-Aminolevulinic acid LC-ESI-QQ (API3000, Applied Biosystems) 10V, Positive-QTOF | splash10-01q9-1900000000-a5686c059e357bc14e96 | 2012-08-31 | HMDB team, MONA | View Spectrum | | Experimental LC-MS/MS | LC-MS/MS Spectrum - 5-Aminolevulinic acid LC-ESI-QQ (API3000, Applied Biosystems) 20V, Positive-QTOF | splash10-000i-9300000000-8e219c18bb0fd0837d82 | 2012-08-31 | HMDB team, MONA | View Spectrum | | Experimental LC-MS/MS | LC-MS/MS Spectrum - 5-Aminolevulinic acid LC-ESI-QQ (API3000, Applied Biosystems) 30V, Positive-QTOF | splash10-0avr-9000000000-b0d0d9b25a36e5c49f2a | 2012-08-31 | HMDB team, MONA | View Spectrum | | Experimental LC-MS/MS | LC-MS/MS Spectrum - 5-Aminolevulinic acid LC-ESI-QQ (API3000, Applied Biosystems) 40V, Positive-QTOF | splash10-0a4i-9000000000-96cc61ad07db2c38c952 | 2012-08-31 | HMDB team, MONA | View Spectrum | | Experimental LC-MS/MS | LC-MS/MS Spectrum - 5-Aminolevulinic acid LC-ESI-QQ (API3000, Applied Biosystems) 50V, Positive-QTOF | splash10-0a4i-9000000000-7b8165e702ac7e6d5f34 | 2012-08-31 | HMDB team, MONA | View Spectrum | | Experimental LC-MS/MS | LC-MS/MS Spectrum - 5-Aminolevulinic acid LC-ESI-QTOF (UPLC Q-Tof Premier, Waters) , Positive-QTOF | splash10-01p9-8900000000-0b739a89ed524e47e3a2 | 2012-08-31 | HMDB team, MONA | View Spectrum | | Experimental LC-MS/MS | LC-MS/MS Spectrum - 5-Aminolevulinic acid LC-ESI-QTOF (UPLC Q-Tof Premier, Waters) , Negative-QTOF | splash10-001i-0900000000-1ffb96adb6268888f722 | 2012-08-31 | HMDB team, MONA | View Spectrum | | Experimental LC-MS/MS | LC-MS/MS Spectrum - 5-Aminolevulinic acid LC-ESI-QTOF , negative-QTOF | splash10-001i-0900000000-1ffb96adb6268888f722 | 2017-09-14 | HMDB team, MONA | View Spectrum | | Experimental LC-MS/MS | LC-MS/MS Spectrum - 5-Aminolevulinic acid LC-ESI-QQ , positive-QTOF | splash10-01q9-1900000000-a5686c059e357bc14e96 | 2017-09-14 | HMDB team, MONA | View Spectrum | | Experimental LC-MS/MS | LC-MS/MS Spectrum - 5-Aminolevulinic acid LC-ESI-QQ , positive-QTOF | splash10-000i-9300000000-8e219c18bb0fd0837d82 | 2017-09-14 | HMDB team, MONA | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 5-Aminolevulinic acid 10V, Positive-QTOF | splash10-03di-4900000000-1669ed2d77c2a1921a2d | 2015-05-26 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 5-Aminolevulinic acid 20V, Positive-QTOF | splash10-0292-9300000000-c07de16859b85d8fb54c | 2015-05-26 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 5-Aminolevulinic acid 40V, Positive-QTOF | splash10-067j-9000000000-316eb66f80f8b40f4ae2 | 2015-05-26 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 5-Aminolevulinic acid 10V, Positive-QTOF | splash10-03di-4900000000-1669ed2d77c2a1921a2d | 2015-05-26 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 5-Aminolevulinic acid 20V, Positive-QTOF | splash10-0292-9300000000-c07de16859b85d8fb54c | 2015-05-26 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 5-Aminolevulinic acid 40V, Positive-QTOF | splash10-067j-9000000000-316eb66f80f8b40f4ae2 | 2015-05-26 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 5-Aminolevulinic acid 10V, Negative-QTOF | splash10-001i-2900000000-e2f3aecb5b3f533e4905 | 2015-05-27 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 5-Aminolevulinic acid 20V, Negative-QTOF | splash10-0bu0-9600000000-fc4f9eaeca47b90745ae | 2015-05-27 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 5-Aminolevulinic acid 40V, Negative-QTOF | splash10-0a4l-9000000000-304beef010b4b4fdbb53 | 2015-05-27 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 5-Aminolevulinic acid 10V, Negative-QTOF | splash10-001i-2900000000-e2f3aecb5b3f533e4905 | 2015-05-27 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 5-Aminolevulinic acid 20V, Negative-QTOF | splash10-0bu0-9600000000-fc4f9eaeca47b90745ae | 2015-05-27 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 5-Aminolevulinic acid 40V, Negative-QTOF | splash10-0a4l-9000000000-304beef010b4b4fdbb53 | 2015-05-27 | Wishart Lab | View Spectrum |
NMR Spectra| Spectrum Type | Description | Deposition Date | Source | View |
|---|
| Experimental 1D NMR | 1H NMR Spectrum (1D, 500 MHz, H2O, experimental) | 2012-12-04 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 1H NMR Spectrum (1D, 100 MHz, D2O, predicted) | 2021-09-29 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 13C NMR Spectrum (1D, 100 MHz, D2O, predicted) | 2021-09-29 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 1H NMR Spectrum (1D, 1000 MHz, D2O, predicted) | 2021-09-29 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 13C NMR Spectrum (1D, 1000 MHz, D2O, predicted) | 2021-09-29 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 1H NMR Spectrum (1D, 200 MHz, D2O, predicted) | 2021-09-29 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 13C NMR Spectrum (1D, 200 MHz, D2O, predicted) | 2021-09-29 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 1H NMR Spectrum (1D, 300 MHz, D2O, predicted) | 2021-09-29 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 13C NMR Spectrum (1D, 300 MHz, D2O, predicted) | 2021-09-29 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 1H NMR Spectrum (1D, 400 MHz, D2O, predicted) | 2021-09-29 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 13C NMR Spectrum (1D, 400 MHz, D2O, predicted) | 2021-09-29 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 1H NMR Spectrum (1D, 500 MHz, D2O, predicted) | 2021-09-29 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 13C NMR Spectrum (1D, 500 MHz, D2O, predicted) | 2021-09-29 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 1H NMR Spectrum (1D, 600 MHz, D2O, predicted) | 2021-09-29 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 13C NMR Spectrum (1D, 600 MHz, D2O, predicted) | 2021-09-29 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 1H NMR Spectrum (1D, 700 MHz, D2O, predicted) | 2021-09-29 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 13C NMR Spectrum (1D, 700 MHz, D2O, predicted) | 2021-09-29 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 1H NMR Spectrum (1D, 800 MHz, D2O, predicted) | 2021-09-29 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 13C NMR Spectrum (1D, 800 MHz, D2O, predicted) | 2021-09-29 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 1H NMR Spectrum (1D, 900 MHz, D2O, predicted) | 2021-09-29 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 13C NMR Spectrum (1D, 900 MHz, D2O, predicted) | 2021-09-29 | Wishart Lab | View Spectrum | | Experimental 2D NMR | [1H, 13C]-HSQC NMR Spectrum (2D, 600 MHz, H2O, experimental) | 2012-12-05 | Wishart Lab | View Spectrum |
IR Spectra| Spectrum Type | Description | Deposition Date | Source | View |
|---|
| Predicted IR Spectrum | IR Ion Spectrum (Predicted IRIS Spectrum, Adduct: [M-H]-) | 2023-02-03 | FELIX lab | View Spectrum | | Predicted IR Spectrum | IR Ion Spectrum (Predicted IRIS Spectrum, Adduct: [M+H]+) | 2023-02-03 | FELIX lab | View Spectrum | | Predicted IR Spectrum | IR Ion Spectrum (Predicted IRIS Spectrum, Adduct: [M+Na]+) | 2023-02-03 | FELIX lab | View Spectrum |
|
|---|
| General References | - Murata K, Sakai T, Morita Y, Iwata T, Dakeishi M: Critical dose of lead affecting delta-aminolevulinic acid levels. J Occup Health. 2003 Jul;45(4):209-14. [PubMed:14646278 ]
- Lu L, Lin G, Xu M, Zou H, Wang Q: [Re-assessment of indicators for screening lead poisoning]. Zhonghua Yu Fang Yi Xue Za Zhi. 1999 Sep;33(5):275-8. [PubMed:11864490 ]
- Landry JL, Gelet A, Bouvier R, Dubernard JM, Martin X, Colombel M: Detection of bladder dysplasia using 5-aminolaevulinic acid-induced porphyrin fluorescence. BJU Int. 2003 May;91(7):623-6. [PubMed:12699472 ]
- Sassa S: Diagnosis and therapy of acute intermittent porphyria. Blood Rev. 1996 Mar;10(1):53-8. [PubMed:8861279 ]
- Tauber S, Stepp H, Meier R, Bone A, Hofstetter A, Stief C: Integral spectrophotometric analysis of 5-aminolaevulinic acid-induced fluorescence cytology of the urinary bladder. BJU Int. 2006 May;97(5):992-6. [PubMed:16643481 ]
- Bhardwaj RK, Herrera-Ruiz D, Sinko PJ, Gudmundsson OS, Knipp G: Delineation of human peptide transporter 1 (hPepT1)-mediated uptake and transport of substrates with varying transporter affinities utilizing stably transfected hPepT1/Madin-Darby canine kidney clones and Caco-2 cells. J Pharmacol Exp Ther. 2005 Sep;314(3):1093-100. Epub 2005 May 18. [PubMed:15901802 ]
- Tschudy DP, Valsamis M, Magnussen CR: Acute intermittent porphyria: clinical and selected research aspects. Ann Intern Med. 1975 Dec;83(6):851-64. [PubMed:1106284 ]
- Lipinski M, Jeromin L: Comparison of the bladder tumour antigen test with photodynamic diagnosis in patients with pathologically confirmed recurrent superficial urinary bladder tumours. BJU Int. 2002 May;89(7):757-9. [PubMed:11966640 ]
- Zareba G, Chmielnicka J: Disturbances in heme biosynthesis in rabbits after administration per os of low doses of tin or lead. Biol Trace Elem Res. 1992 Aug;34(2):115-22. [PubMed:1381933 ]
- Srivastava G, Borthwick IA, Maguire DJ, Elferink CJ, Bawden MJ, Mercer JF, May BK: Regulation of 5-aminolevulinate synthase mRNA in different rat tissues. J Biol Chem. 1988 Apr 15;263(11):5202-9. [PubMed:3356687 ]
- Maines MD, Mayer RD: Inhibition of testicular cytochrome P-450-dependent steroid biosynthesis by cis-platinum. Reversal by human chorionic gonadotropin. J Biol Chem. 1985 May 25;260(10):6063-8. [PubMed:4039724 ]
- Santos MA, Belo VG, Santos G: Effectiveness of photodynamic therapy with topical 5-aminolevulinic acid and intense pulsed light versus intense pulsed light alone in the treatment of acne vulgaris: comparative study. Dermatol Surg. 2005 Aug;31(8 Pt 1):910-5. [PubMed:16042935 ]
- Gederaas OA, Rasch MH, Berg K, Lagerberg JW, Dubbelman TM: Photodynamically induced effects in colon carcinoma cells (WiDr) by endogenous photosensitizers generated by incubation with 5-aminolaevulinic acid. J Photochem Photobiol B. 1999 Apr;49(2-3):162-70. [PubMed:10392465 ]
- Lee S, Kollias N, McAuliffe DJ, Flotte TJ, Doukas AG: Topical drug delivery in humans with a single photomechanical wave. Pharm Res. 1999 Nov;16(11):1717-21. [PubMed:10571277 ]
- van den Akker JT, Boot K, Vernon DI, Brown SB, Groenendijk L, van Rhoon GC, Sterenborg HJ: Effect of elevating the skin temperature during topical ALA application on in vitro ALA penetration through mouse skin and in vivo PpIX production in human skin. Photochem Photobiol Sci. 2004 Mar;3(3):263-7. Epub 2004 Feb 13. [PubMed:14993942 ]
- Lerda D: Study of sperm characteristics in persons occupationally exposed to lead. Am J Ind Med. 1992;22(4):567-71. [PubMed:1442789 ]
- Winkler A, Muller-Goymann CC: Comparative permeation studies for delta-aminolevulinic acid and its n-butylester through stratum corneum and artificial skin constructs. Eur J Pharm Biopharm. 2002 May;53(3):281-7. [PubMed:11976015 ]
- Authors unspecified: Hexyl aminolevulinate: 5-ALA hexylester, 5-ALA hexylesther, aminolevulinic acid hexyl ester, hexaminolevulinate, hexyl 5-aminolevulinate, P 1206. Drugs R D. 2005;6(4):235-8. [PubMed:15991884 ]
- Collaud S, Jichlinski P, Marti A, Aymon D, Gurny R, Lange N: An open pharmacokinetic study of hexylaminolevulinate-induced photodiagnosis after intravesical administration. Drugs R D. 2006;7(3):173-86. [PubMed:16752943 ]
- Fritsch C, Batz J, Bolsen K, Schulte KW, Zumdick M, Ruzicka T, Goerz G: Ex vivo application of delta-aminolevulinic acid induces high and specific porphyrin levels in human skin tumors: possible basis for selective photodynamic therapy. Photochem Photobiol. 1997 Jul;66(1):114-8. [PubMed:9230710 ]
|
|---|