| Record Information |
|---|
| Version | 5.0 |
|---|
| Status | Expected but not Quantified |
|---|
| Creation Date | 2005-11-16 15:48:42 UTC |
|---|
| Update Date | 2021-09-14 14:59:35 UTC |
|---|
| HMDB ID | HMDB0001162 |
|---|
| Secondary Accession Numbers | |
|---|
| Metabolite Identification |
|---|
| Common Name | Heme O |
|---|
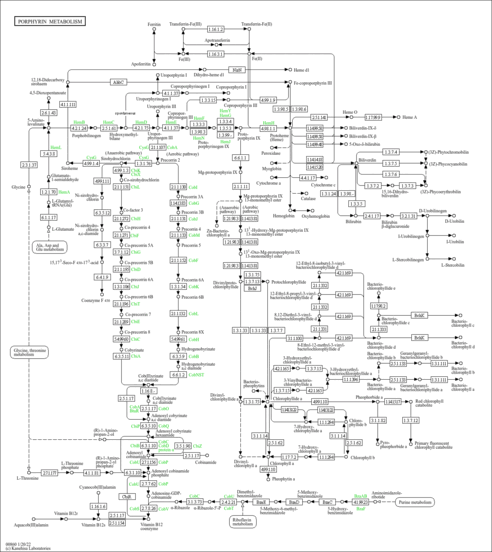
| Description | Heme O belongs to the class of organic compounds known as metallotetrapyrroles. These are polycyclic compounds containing a tetrapyrrole skeleton combined with a metal atom. Heme O exists in all living species, ranging from bacteria to plants to humans. In humans, heme O is involved in the metabolic disorder called the congenital erythropoietic porphyria (cep) or gunther disease pathway. Heme O has been detected, but not quantified in, a few different foods, such as anatidaes (Anatidae), chickens (Gallus gallus), and domestic pigs (Sus scrofa domestica). This could make heme O a potential biomarker for the consumption of these foods. Heme O is a primary metabolite. Primary metabolites are metabolically or physiologically essential metabolites. They are directly involved in an organism’s growth, development or reproduction. Based on a literature review a significant number of articles have been published on Heme O. |
|---|
| Structure | CC(C)=CCC\C(C)=C\CC\C(C)=C\CCC(O)C1=C(C)/C2=C/C3=N/C(=C\C4=C(CCC(O)=O)C(C)=C5/C=C6\N=C(\C=C\1/N\2[Fe]N45)C(C)=C6C=C)/C(CCC(O)=O)=C3C InChI=1S/C49H60N4O5.Fe/c1-10-35-31(6)40-26-45-49(46(54)19-13-18-30(5)17-12-16-29(4)15-11-14-28(2)3)34(9)41(53-45)24-38-32(7)36(20-22-47(55)56)43(51-38)27-44-37(21-23-48(57)58)33(8)39(52-44)25-42(35)50-40;/h10,14,16,18,24-27,46,54H,1,11-13,15,17,19-23H2,2-9H3,(H4,50,51,52,53,55,56,57,58);/q;+2/p-2/b29-16+,30-18+,38-24-,39-25-,40-26-,41-24-,42-25-,43-27-,44-27-,45-26-; |
|---|
| Synonyms | Not Available |
|---|
| Chemical Formula | C49H58FeN4O5 |
|---|
| Average Molecular Weight | 838.854 |
|---|
| Monoisotopic Molecular Weight | 838.375663119 |
|---|
| IUPAC Name | 3-[(11Z,16Z)-20-(2-carboxyethyl)-15-ethenyl-10-[(4E,8E)-1-hydroxy-5,9,13-trimethyltetradeca-4,8,12-trien-1-yl]-5,9,14,19-tetramethyl-21,23,24,25-tetraaza-22-ferrahexacyclo[9.9.3.1³,⁶.1¹³,¹⁶.0⁸,²³.0¹⁸,²¹]pentacosa-1(20),2,4,6(25),7,9,11,13(24),14,16,18-undecaen-4-yl]propanoic acid |
|---|
| Traditional Name | 3-[(11Z,16Z)-20-(2-carboxyethyl)-15-ethenyl-10-[(4E,8E)-1-hydroxy-5,9,13-trimethyltetradeca-4,8,12-trien-1-yl]-5,9,14,19-tetramethyl-21,23,24,25-tetraaza-22-ferrahexacyclo[9.9.3.1³,⁶.1¹³,¹⁶.0⁸,²³.0¹⁸,²¹]pentacosa-1(20),2,4,6(25),7,9,11,13(24),14,16,18-undecaen-4-yl]propanoic acid |
|---|
| CAS Registry Number | 137397-56-9 |
|---|
| SMILES | CC(C)=CCC\C(C)=C\CC\C(C)=C\CCC(O)C1=C(C)/C2=C/C3=N/C(=C\C4=C(CCC(O)=O)C(C)=C5/C=C6\N=C(\C=C\1/N\2[Fe]N45)C(C)=C6C=C)/C(CCC(O)=O)=C3C |
|---|
| InChI Identifier | InChI=1S/C49H60N4O5.Fe/c1-10-35-31(6)40-26-45-49(46(54)19-13-18-30(5)17-12-16-29(4)15-11-14-28(2)3)34(9)41(53-45)24-38-32(7)36(20-22-47(55)56)43(51-38)27-44-37(21-23-48(57)58)33(8)39(52-44)25-42(35)50-40;/h10,14,16,18,24-27,46,54H,1,11-13,15,17,19-23H2,2-9H3,(H4,50,51,52,53,55,56,57,58);/q;+2/p-2/b29-16+,30-18+,38-24-,39-25-,40-26-,41-24-,42-25-,43-27-,44-27-,45-26-; |
|---|
| InChI Key | FISPASSVCDRERW-KVGORYHISA-L |
|---|
| Chemical Taxonomy |
|---|
| Description | Belongs to the class of organic compounds known as metallotetrapyrroles. These are polycyclic compounds containing a tetrapyrrole skeleton combined with a metal atom. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Organoheterocyclic compounds |
|---|
| Class | Tetrapyrroles and derivatives |
|---|
| Sub Class | Metallotetrapyrroles |
|---|
| Direct Parent | Metallotetrapyrroles |
|---|
| Alternative Parents | Not Available |
|---|
| Substituents | Not Available |
|---|
| Molecular Framework | Not Available |
|---|
| External Descriptors | Not Available |
|---|
| Ontology |
|---|
| Physiological effect | Not Available |
|---|
| Disposition | |
|---|
| Process | |
|---|
| Role | Not Available |
|---|
| Physical Properties |
|---|
| State | Solid |
|---|
| Experimental Molecular Properties | | Property | Value | Reference |
|---|
| Melting Point | Not Available | Not Available | | Boiling Point | Not Available | Not Available | | Water Solubility | Not Available | Not Available | | LogP | Not Available | Not Available |
|
|---|
| Experimental Chromatographic Properties | Not Available |
|---|
| Predicted Molecular Properties | |
|---|
| Predicted Chromatographic Properties | Predicted Collision Cross SectionsPredicted Retention Times Underivatized| Chromatographic Method | Retention Time | Reference |
|---|
| AjsUoB = Accucore 150 Amide HILIC with 10mM Ammonium Formate, 0.1% Formic Acid | 70.8 seconds | 40023050 | | Fem_Long = Waters ACQUITY UPLC HSS T3 C18 with Water:MeOH and 0.1% Formic Acid | 4646.1 seconds | 40023050 | | Fem_Lipids = Ascentis Express C18 with (60:40 water:ACN):(90:10 IPA:ACN) and 10mM NH4COOH + 0.1% Formic Acid | 186.7 seconds | 40023050 | | Life_Old = Waters ACQUITY UPLC BEH C18 with Water:(20:80 acetone:ACN) and 0.1% Formic Acid | 305.3 seconds | 40023050 | | Life_New = RP Waters ACQUITY UPLC HSS T3 C18 with Water:(30:70 MeOH:ACN) and 0.1% Formic Acid | 171.5 seconds | 40023050 | | RIKEN = Waters ACQUITY UPLC BEH C18 with Water:ACN and 0.1% Formic Acid | 153.3 seconds | 40023050 | | Eawag_XBridgeC18 = XBridge C18 3.5u 2.1x50 mm with Water:MeOH and 0.1% Formic Acid | 923.6 seconds | 40023050 | | BfG_NTS_RP1 =Agilent Zorbax Eclipse Plus C18 (2.1 mm x 150 mm, 3.5 um) with Water:ACN and 0.1% Formic Acid | 968.5 seconds | 40023050 | | HILIC_BDD_2 = Merck SeQuant ZIC-HILIC with ACN(0.1% formic acid):water(16 mM ammonium formate) | 113.6 seconds | 40023050 | | UniToyama_Atlantis = RP Waters Atlantis T3 (2.1 x 150 mm, 5 um) with ACN:Water and 0.1% Formic Acid | 1915.4 seconds | 40023050 | | BDD_C18 = Hypersil Gold 1.9µm C18 with Water:ACN and 0.1% Formic Acid | 832.9 seconds | 40023050 | | UFZ_Phenomenex = Kinetex Core-Shell C18 2.6 um, 3.0 x 100 mm, Phenomenex with Water:MeOH and 0.1% Formic Acid | 2051.3 seconds | 40023050 | | SNU_RIKEN_POS = Waters ACQUITY UPLC BEH C18 with Water:ACN and 0.1% Formic Acid | 618.9 seconds | 40023050 | | RPMMFDA = Waters ACQUITY UPLC BEH C18 with Water:ACN and 0.1% Formic Acid | 558.6 seconds | 40023050 | | MTBLS87 = Merck SeQuant ZIC-pHILIC column with ACN:Water and :ammonium carbonate | 154.0 seconds | 40023050 | | KI_GIAR_zic_HILIC_pH2_7 = Merck SeQuant ZIC-HILIC with ACN:Water and 0.1% FA | 423.9 seconds | 40023050 | | Meister zic-pHILIC pH9.3 = Merck SeQuant ZIC-pHILIC column with ACN:Water 5mM NH4Ac pH9.3 and 5mM ammonium acetate in water | 9.8 seconds | 40023050 |
Predicted Kovats Retention IndicesUnderivatized |
|---|
| MS/MS Spectra| Spectrum Type | Description | Splash Key | Deposition Date | Source | View |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - Heme O 10V, Positive-QTOF | splash10-009l-0000000970-0ab5b5c8236aeb83353a | 2015-09-14 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - Heme O 20V, Positive-QTOF | splash10-00s6-1200001930-2056d7e5ba2335a6154b | 2015-09-14 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - Heme O 40V, Positive-QTOF | splash10-0159-1100000900-fb1e1bfe680d13bc4e01 | 2015-09-14 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - Heme O 10V, Negative-QTOF | splash10-000i-0000000390-fc9d0bdfd86ce5bb111e | 2015-09-15 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - Heme O 20V, Negative-QTOF | splash10-0171-2120061950-c114e5cb2af8c6ea5b24 | 2015-09-15 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - Heme O 40V, Negative-QTOF | splash10-00fs-0000090410-19e8eef5041c9549ea3f | 2015-09-15 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - Heme O 10V, Negative-QTOF | splash10-000i-0000000090-46c44cfe9d493547a919 | 2021-09-21 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - Heme O 20V, Negative-QTOF | splash10-00ri-0100002790-aa7b2dbeb177907521b7 | 2021-09-21 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - Heme O 40V, Negative-QTOF | splash10-00g0-0000024930-47277d8050776dbaab1c | 2021-09-21 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - Heme O 10V, Positive-QTOF | splash10-0079-2000001690-d482b755c66f4c96c8d0 | 2021-09-25 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - Heme O 20V, Positive-QTOF | splash10-003s-6200006910-8a7a86584f45027cc939 | 2021-09-25 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - Heme O 40V, Positive-QTOF | splash10-067j-8400009320-337b466a95de04a44a14 | 2021-09-25 | Wishart Lab | View Spectrum |
NMR Spectra| Spectrum Type | Description | Deposition Date | Source | View |
|---|
| Predicted 1D NMR | 13C NMR Spectrum (1D, 100 MHz, H2O, predicted) | 2022-08-17 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 1H NMR Spectrum (1D, 100 MHz, H2O, predicted) | 2022-08-17 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 13C NMR Spectrum (1D, 1000 MHz, H2O, predicted) | 2022-08-17 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 1H NMR Spectrum (1D, 1000 MHz, H2O, predicted) | 2022-08-17 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 13C NMR Spectrum (1D, 200 MHz, H2O, predicted) | 2022-08-17 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 1H NMR Spectrum (1D, 200 MHz, H2O, predicted) | 2022-08-17 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 13C NMR Spectrum (1D, 300 MHz, H2O, predicted) | 2022-08-17 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 1H NMR Spectrum (1D, 300 MHz, H2O, predicted) | 2022-08-17 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 13C NMR Spectrum (1D, 400 MHz, H2O, predicted) | 2022-08-17 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 1H NMR Spectrum (1D, 400 MHz, H2O, predicted) | 2022-08-17 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 13C NMR Spectrum (1D, 500 MHz, H2O, predicted) | 2022-08-17 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 1H NMR Spectrum (1D, 500 MHz, H2O, predicted) | 2022-08-17 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 13C NMR Spectrum (1D, 600 MHz, H2O, predicted) | 2022-08-17 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 1H NMR Spectrum (1D, 600 MHz, H2O, predicted) | 2022-08-17 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 13C NMR Spectrum (1D, 700 MHz, H2O, predicted) | 2022-08-17 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 1H NMR Spectrum (1D, 700 MHz, H2O, predicted) | 2022-08-17 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 13C NMR Spectrum (1D, 800 MHz, H2O, predicted) | 2022-08-17 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 1H NMR Spectrum (1D, 800 MHz, H2O, predicted) | 2022-08-17 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 13C NMR Spectrum (1D, 900 MHz, H2O, predicted) | 2022-08-17 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 1H NMR Spectrum (1D, 900 MHz, H2O, predicted) | 2022-08-17 | Wishart Lab | View Spectrum |
|
|---|