| Record Information |
|---|
| Version | 5.0 |
|---|
| Status | Expected but not Quantified |
|---|
| Creation Date | 2005-11-16 15:48:42 UTC |
|---|
| Update Date | 2021-09-14 15:47:20 UTC |
|---|
| HMDB ID | HMDB0001179 |
|---|
| Secondary Accession Numbers | |
|---|
| Metabolite Identification |
|---|
| Common Name | Nicotinic acid adenine dinucleotide |
|---|
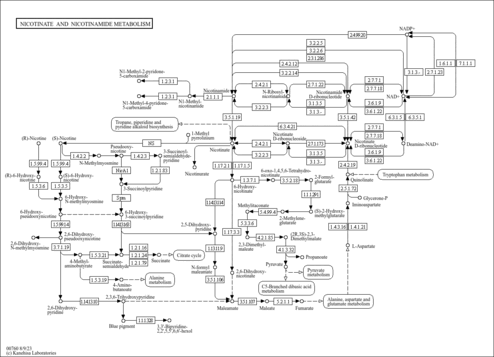
| Description | Nicotinic acid adenine dinucleotide, also known as deamido-NAD or NAAD, belongs to the class of organic compounds known as (5'->5')-dinucleotides. These are dinucleotides where the two bases are connected via a (5'->5')-phosphodiester linkage. NAAD is possibly soluble (in water) and a strong basic compound (based on its pKa). NAAD exists in all living species, ranging from bacteria to humans. L-Glutamine and NAAD can be converted into L-glutamic acid and NAD; which is catalyzed by the enzyme glutamine-dependent nad(+) synthetase. In humans, NAAD is involved in the nicotinate and nicotinamide metabolism pathway. NAAD is also involved in the metabolic disorder called succinic semialdehyde dehydrogenase deficiency. Outside of the human body, NAAD has been detected, but not quantified in, several different foods, such as japanese walnuts, cauliflowers, sparkleberries, komatsuna, and macadamia nut (m. tetraphylla). This could make NAAD a potential biomarker for the consumption of these foods. NAAD is the product of the degradation of Nicotinic acid adenine dinucleotide phosphate (NAADP) by a Ca2+-sensitive phosphatase. NAADP is a Ca2+-mobilizing second messenger which is synthesized, in response to extracellular stimuli, via the base-exchange reaction by an ADP-ribosyl cyclase (ARC) family members (such as CD38). NAADP binds to and opens Ca2+ channels on intracellular organelles, thereby increasing the intracellular Ca2+ concentration which, in turn, modulates a variety of cellular processes. Structurally, NAADP it is a dinucleotide that only differs from the house-keeping enzyme cofactor, NADP, by a hydroxyl group (replacing the nicotinamide amino group) and yet this minor modification converts it into the most potent Ca2+-mobilizing second messenger yet described. NAADP may also be broken down to 2-phosphoadenosine diphosphoribose (ADPRP) by CD38 or reduced to NAADPH. |
|---|
| Structure | NC1=C2N=CN([C@@H]3O[C@H](COP(O)(=O)OP(O)(=O)OC[C@H]4O[C@H]([C@H](O)[C@@H]4O)[N+]4=CC=CC(=C4)C(O)=O)[C@@H](O)[C@H]3O)C2=NC=N1 InChI=1S/C21H26N6O15P2/c22-17-12-18(24-7-23-17)27(8-25-12)20-16(31)14(29)11(41-20)6-39-44(36,37)42-43(34,35)38-5-10-13(28)15(30)19(40-10)26-3-1-2-9(4-26)21(32)33/h1-4,7-8,10-11,13-16,19-20,28-31H,5-6H2,(H4-,22,23,24,32,33,34,35,36,37)/p+1/t10-,11-,13-,14-,15-,16-,19-,20-/m1/s1 |
|---|
| Synonyms | | Value | Source |
|---|
| Deamido-NAD | ChEBI | | Deamido-nad+ | ChEBI | | Deamino-nad+ | ChEBI | | Nicotinate adenine dinucleotide | Generator | | Deamidonicotinamide adenine dinucleoetide | HMDB | | NAAD | HMDB | | NaADN | HMDB | | Adenine-nicotinic acid dinucleotide | HMDB | | Deamidodiphosphopyridine nucleotide | HMDB | | Desamido NAD | HMDB | | Nicotinic acid-adenine dinucleotide | HMDB |
|
|---|
| Chemical Formula | C21H27N6O15P2 |
|---|
| Average Molecular Weight | 665.4178 |
|---|
| Monoisotopic Molecular Weight | 665.100962248 |
|---|
| IUPAC Name | 1-[(2R,3R,4S,5R)-5-[({[({[(2R,3S,4R,5R)-5-(6-amino-9H-purin-9-yl)-3,4-dihydroxyoxolan-2-yl]methoxy}(hydroxy)phosphoryl)oxy](hydroxy)phosphoryl}oxy)methyl]-3,4-dihydroxyoxolan-2-yl]-3-carboxy-1lambda5-pyridin-1-ylium |
|---|
| Traditional Name | 1-[(2R,3R,4S,5R)-5-{[({[(2R,3S,4R,5R)-5-(6-aminopurin-9-yl)-3,4-dihydroxyoxolan-2-yl]methoxy(hydroxy)phosphoryl}oxy(hydroxy)phosphoryl)oxy]methyl}-3,4-dihydroxyoxolan-2-yl]-3-carboxy-1lambda5-pyridin-1-ylium |
|---|
| CAS Registry Number | 6450-77-7 |
|---|
| SMILES | NC1=C2N=CN([C@@H]3O[C@H](COP(O)(=O)OP(O)(=O)OC[C@H]4O[C@H]([C@H](O)[C@@H]4O)[N+]4=CC=CC(=C4)C(O)=O)[C@@H](O)[C@H]3O)C2=NC=N1 |
|---|
| InChI Identifier | InChI=1S/C21H26N6O15P2/c22-17-12-18(24-7-23-17)27(8-25-12)20-16(31)14(29)11(41-20)6-39-44(36,37)42-43(34,35)38-5-10-13(28)15(30)19(40-10)26-3-1-2-9(4-26)21(32)33/h1-4,7-8,10-11,13-16,19-20,28-31H,5-6H2,(H4-,22,23,24,32,33,34,35,36,37)/p+1/t10-,11-,13-,14-,15-,16-,19-,20-/m1/s1 |
|---|
| InChI Key | SENPVEZBRZQVST-HISDBWNOSA-O |
|---|
| Chemical Taxonomy |
|---|
| Description | Belongs to the class of organic compounds known as (5'->5')-dinucleotides. These are dinucleotides where the two bases are connected via a (5'->5')-phosphodiester linkage. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Nucleosides, nucleotides, and analogues |
|---|
| Class | (5'->5')-dinucleotides |
|---|
| Sub Class | Not Available |
|---|
| Direct Parent | (5'->5')-dinucleotides |
|---|
| Alternative Parents | |
|---|
| Substituents | - (5'->5')-dinucleotide
- Purine nucleotide sugar
- Purine ribonucleoside diphosphate
- Purine ribonucleoside monophosphate
- Nicotinic-acid-nucleotide
- Pyridine nucleotide
- Pentose phosphate
- Pentose-5-phosphate
- Glycosyl compound
- N-glycosyl compound
- 6-aminopurine
- Monosaccharide phosphate
- Organic pyrophosphate
- Imidazopyrimidine
- Purine
- Pyridine carboxylic acid or derivatives
- Pyridine carboxylic acid
- Aminopyrimidine
- Monoalkyl phosphate
- Pyridinium
- Pyridine
- Organic phosphoric acid derivative
- Alkyl phosphate
- N-substituted imidazole
- Pyrimidine
- Imidolactam
- Monosaccharide
- Phosphoric acid ester
- Tetrahydrofuran
- Heteroaromatic compound
- Azole
- Imidazole
- Vinylogous amide
- Amino acid
- Secondary alcohol
- Amino acid or derivatives
- Carboxylic acid
- Carboxylic acid derivative
- Oxacycle
- Azacycle
- Monocarboxylic acid or derivatives
- Organoheterocyclic compound
- Alcohol
- Amine
- Organic oxide
- Organopnictogen compound
- Organic oxygen compound
- Hydrocarbon derivative
- Organonitrogen compound
- Organooxygen compound
- Organic nitrogen compound
- Primary amine
- Organic cation
- Aromatic heteropolycyclic compound
|
|---|
| Molecular Framework | Aromatic heteropolycyclic compounds |
|---|
| External Descriptors | |
|---|
| Ontology |
|---|
| Not Available | Not Available |
|---|
| Physical Properties |
|---|
| State | Solid |
|---|
| Experimental Molecular Properties | | Property | Value | Reference |
|---|
| Melting Point | Not Available | Not Available | | Boiling Point | Not Available | Not Available | | Water Solubility | Not Available | Not Available | | LogP | Not Available | Not Available |
|
|---|
| Experimental Chromatographic Properties | Not Available |
|---|
| Predicted Molecular Properties | |
|---|
| Predicted Chromatographic Properties | Predicted Collision Cross SectionsPredicted Kovats Retention IndicesUnderivatized |
|---|
| GC-MS Spectra| Spectrum Type | Description | Splash Key | Deposition Date | Source | View |
|---|
| Predicted GC-MS | Predicted GC-MS Spectrum - Nicotinic acid adenine dinucleotide GC-MS (Non-derivatized) - 70eV, Positive | splash10-0002-1202903000-23becb537df72d0e35b9 | 2017-09-01 | Wishart Lab | View Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - Nicotinic acid adenine dinucleotide GC-MS (TMS_1_1) - 70eV, Positive | Not Available | 2021-10-18 | Wishart Lab | View Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - Nicotinic acid adenine dinucleotide GC-MS (TMS_1_2) - 70eV, Positive | Not Available | 2021-10-18 | Wishart Lab | View Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - Nicotinic acid adenine dinucleotide GC-MS (TMS_1_3) - 70eV, Positive | Not Available | 2021-10-18 | Wishart Lab | View Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - Nicotinic acid adenine dinucleotide GC-MS (TMS_1_4) - 70eV, Positive | Not Available | 2021-10-18 | Wishart Lab | View Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - Nicotinic acid adenine dinucleotide GC-MS (TMS_1_5) - 70eV, Positive | Not Available | 2021-10-18 | Wishart Lab | View Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - Nicotinic acid adenine dinucleotide GC-MS (TMS_1_6) - 70eV, Positive | Not Available | 2021-10-18 | Wishart Lab | View Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - Nicotinic acid adenine dinucleotide GC-MS (TMS_1_7) - 70eV, Positive | Not Available | 2021-10-18 | Wishart Lab | View Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - Nicotinic acid adenine dinucleotide GC-MS (TMS_1_8) - 70eV, Positive | Not Available | 2021-10-18 | Wishart Lab | View Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - Nicotinic acid adenine dinucleotide GC-MS (TMS_2_1) - 70eV, Positive | Not Available | 2021-10-18 | Wishart Lab | View Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - Nicotinic acid adenine dinucleotide GC-MS (TMS_2_2) - 70eV, Positive | Not Available | 2021-10-18 | Wishart Lab | View Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - Nicotinic acid adenine dinucleotide GC-MS (TMS_2_3) - 70eV, Positive | Not Available | 2021-10-18 | Wishart Lab | View Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - Nicotinic acid adenine dinucleotide GC-MS (TMS_2_4) - 70eV, Positive | Not Available | 2021-10-18 | Wishart Lab | View Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - Nicotinic acid adenine dinucleotide GC-MS (TMS_2_5) - 70eV, Positive | Not Available | 2021-10-18 | Wishart Lab | View Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - Nicotinic acid adenine dinucleotide GC-MS (TMS_2_6) - 70eV, Positive | Not Available | 2021-10-18 | Wishart Lab | View Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - Nicotinic acid adenine dinucleotide GC-MS (TMS_2_7) - 70eV, Positive | Not Available | 2021-10-18 | Wishart Lab | View Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - Nicotinic acid adenine dinucleotide GC-MS (TMS_2_8) - 70eV, Positive | Not Available | 2021-10-18 | Wishart Lab | View Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - Nicotinic acid adenine dinucleotide GC-MS (TMS_2_9) - 70eV, Positive | Not Available | 2021-10-18 | Wishart Lab | View Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - Nicotinic acid adenine dinucleotide GC-MS (TMS_2_10) - 70eV, Positive | Not Available | 2021-10-18 | Wishart Lab | View Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - Nicotinic acid adenine dinucleotide GC-MS (TMS_2_11) - 70eV, Positive | Not Available | 2021-10-18 | Wishart Lab | View Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - Nicotinic acid adenine dinucleotide GC-MS (TMS_2_12) - 70eV, Positive | Not Available | 2021-10-18 | Wishart Lab | View Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - Nicotinic acid adenine dinucleotide GC-MS (TMS_2_13) - 70eV, Positive | Not Available | 2021-10-18 | Wishart Lab | View Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - Nicotinic acid adenine dinucleotide GC-MS (TMS_2_14) - 70eV, Positive | Not Available | 2021-10-18 | Wishart Lab | View Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - Nicotinic acid adenine dinucleotide GC-MS (TMS_2_15) - 70eV, Positive | Not Available | 2021-10-18 | Wishart Lab | View Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - Nicotinic acid adenine dinucleotide GC-MS (TMS_2_16) - 70eV, Positive | Not Available | 2021-10-18 | Wishart Lab | View Spectrum |
MS/MS Spectra| Spectrum Type | Description | Splash Key | Deposition Date | Source | View |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - Nicotinic acid adenine dinucleotide 10V, Positive-QTOF | splash10-014i-0000009000-1ae9f4d0896b777b28c3 | 2015-09-15 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - Nicotinic acid adenine dinucleotide 20V, Positive-QTOF | splash10-014j-0000009000-f1d50bdee2c40a519304 | 2015-09-15 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - Nicotinic acid adenine dinucleotide 40V, Positive-QTOF | splash10-0006-9730204000-6e772ef6b1bb54fbe399 | 2015-09-15 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - Nicotinic acid adenine dinucleotide 10V, Negative-QTOF | splash10-03di-0000009000-a61b90d48d56a74bb6c8 | 2015-09-15 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - Nicotinic acid adenine dinucleotide 20V, Negative-QTOF | splash10-03xr-0100109000-b0e0147bb3637f905fd9 | 2015-09-15 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - Nicotinic acid adenine dinucleotide 40V, Negative-QTOF | splash10-01rt-9300000000-05f34f8e6f2c28916f6d | 2015-09-15 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - Nicotinic acid adenine dinucleotide 10V, Positive-QTOF | splash10-00r6-0100095000-9ad8a85562aab6dafffd | 2021-09-22 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - Nicotinic acid adenine dinucleotide 20V, Positive-QTOF | splash10-00y0-1520194000-13a500ab481c2f4044b7 | 2021-09-22 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - Nicotinic acid adenine dinucleotide 40V, Positive-QTOF | splash10-000i-2911100000-7a5df3987b07b40c3eba | 2021-09-22 | Wishart Lab | View Spectrum |
NMR Spectra| Spectrum Type | Description | Deposition Date | Source | View |
|---|
| Predicted 1D NMR | 1H NMR Spectrum (1D, 100 MHz, D2O, predicted) | 2021-09-16 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 13C NMR Spectrum (1D, 100 MHz, D2O, predicted) | 2021-09-16 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 1H NMR Spectrum (1D, 200 MHz, D2O, predicted) | 2021-09-16 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 13C NMR Spectrum (1D, 200 MHz, D2O, predicted) | 2021-09-16 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 1H NMR Spectrum (1D, 300 MHz, D2O, predicted) | 2021-09-16 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 13C NMR Spectrum (1D, 300 MHz, D2O, predicted) | 2021-09-16 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 1H NMR Spectrum (1D, 400 MHz, D2O, predicted) | 2021-09-16 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 13C NMR Spectrum (1D, 400 MHz, D2O, predicted) | 2021-09-16 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 1H NMR Spectrum (1D, 500 MHz, D2O, predicted) | 2021-09-16 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 13C NMR Spectrum (1D, 500 MHz, D2O, predicted) | 2021-09-16 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 1H NMR Spectrum (1D, 600 MHz, D2O, predicted) | 2021-09-16 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 13C NMR Spectrum (1D, 600 MHz, D2O, predicted) | 2021-09-16 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 1H NMR Spectrum (1D, 700 MHz, D2O, predicted) | 2021-09-16 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 13C NMR Spectrum (1D, 700 MHz, D2O, predicted) | 2021-09-16 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 1H NMR Spectrum (1D, 800 MHz, D2O, predicted) | 2021-09-16 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 13C NMR Spectrum (1D, 800 MHz, D2O, predicted) | 2021-09-16 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 1H NMR Spectrum (1D, 900 MHz, D2O, predicted) | 2021-09-16 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 13C NMR Spectrum (1D, 900 MHz, D2O, predicted) | 2021-09-16 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 1H NMR Spectrum (1D, 1000 MHz, D2O, predicted) | 2021-09-16 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 13C NMR Spectrum (1D, 1000 MHz, D2O, predicted) | 2021-09-16 | Wishart Lab | View Spectrum |
|
|---|